Pancreatic Cystic Lymphangioma Complicated by Haemorrhagic Infarction: A Case Report
CASE REPORT
Hong Kong J Radiol 2025 Mar;28(1):e50-54 | Epub 18 March 2025
Pancreatic Cystic Lymphangioma Complicated by Haemorrhagic Infarction: A Case Report
Patricia Jarmin Lopez Pua1, Florence Los Baños1, Honey Lee Tan2, Victor Tatco2
1 Institute of Radiology, St Luke’s Medical Center, Quezon City, The Philippines
2 Institute of Surgery, St Luke’s Medical Center, Quezon City, The Philippines
Correspondence: Dr PJL Pua, Institute of Radiology, St Luke’s Medical Center, Quezon City, The Philippines. Email: patriciajarmin.pua@gmail.com
Submitted: 16 August 2023; Accepted: 9 February 2024.
Contributors: All authors designed the study, acquired the data, analysed the data, drafted the manuscript, and critically revised the manuscript
for important intellectual content. All authors had full access to the data, contributed to the study, approved the final version for publication, and
take responsibility for its accuracy and integrity.
Conflicts of Interest: All authors have disclosed no conflicts of interest.
Funding/Support: This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data Availability: All data generated or analysed during the present study are available from the corresponding author on reasonable request.
Ethics Approval: The study was approved by the St Luke’s Institutional Ethics Review Committee, the Philippines (Ref No.: SL-23172). Informed consent for the study and publication was obtained from the patient.
INTRODUCTION
Pancreatic cystic lymphangioma or lymphatic
malformation in the pancreas is an extremely rare entity
resulting from lymphatic flow obstruction. It comprises
<1% of all types of lymphatic malformations and about
0.2% of all cystic lesions of the pancreas.[1] Fewer than
100 cases have been reported in the literature.[2] Currently,
there are no guidelines for diagnosis and management.
Preoperative evaluation and management depend on
clinical presentation. Computed tomography (CT),
magnetic resonance imaging (MRI), and ultrasonography
have been widely used for initial assessment but
histopathology remains the standard for diagnostic
confirmation.[3]
We report the case of a patient who presented to the
emergency department with severe right-sided abdominal
pain. Imaging studies revealed a large complex cystic
mass with unclear origin. The patient underwent
surgery that revealed pancreatic cystic lymphangioma
complicated by haemorrhagic infarction.
CASE PRESENTATION
A 43-year-old female presented to the emergency department of our institution in March 2022 with
sudden-onset severe right upper abdominal pain. Bedside
ultrasound in the emergency room revealed a large cystic
abdominal mass. There was no associated jaundice,
vomiting, weight loss or altered bowel movement and
no history of abdominal surgeries. Physical examination
revealed a slightly globular abdomen with epigastric
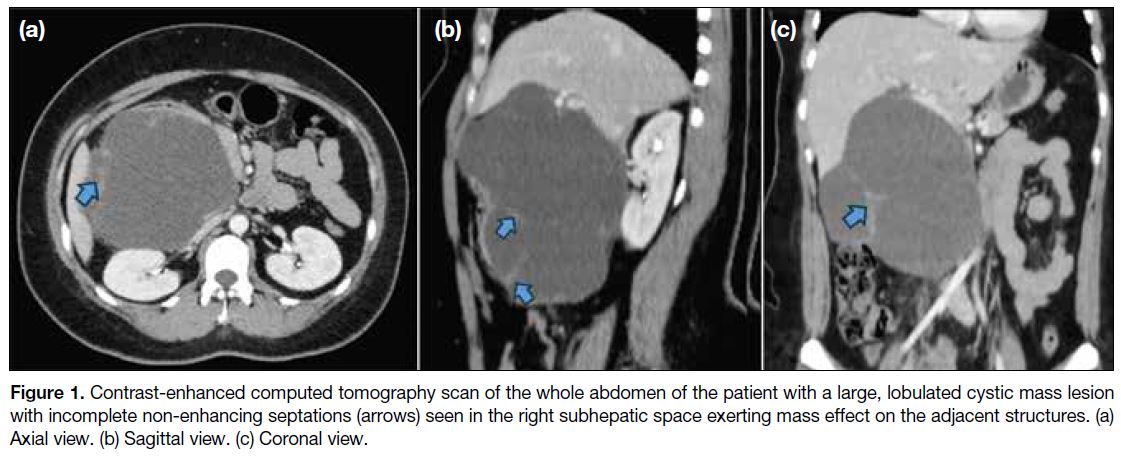
tenderness. Contrast-enhanced CT scan of the abdomen
(Figure 1) showed a lobulated cystic mass with thick
and incomplete minimally enhancing septations
measuring about 10.6 × 13.6 × 17 cm3 (anteroposterior × transverse × craniocaudal). Minimal surrounding fat
stranding densities were seen. The mass was located in
the right subhepatic space, close to the duodenum and
pancreatic head, exerting a mass effect on the liver and
gallbladder, superiorly, and on the right kidney, renal
vessels and inferior vena cava, posteriorly. Marked
luminal narrowing of the inferior vena cava was evident.
There were no enlarged abdominopelvic lymph nodes.
Differential diagnoses were mesenteric cyst, duplication
cyst and cystic neoplasm of the pancreas.
Figure 1. Contrast-enhanced computed tomography scan of the whole abdomen of the patient with a large, lobulated cystic mass lesion
with incomplete non-enhancing septations (arrows) seen in the right subhepatic space exerting mass effect on the adjacent structures. (a)
Axial view. (b) Sagittal view. (c) Coronal view.
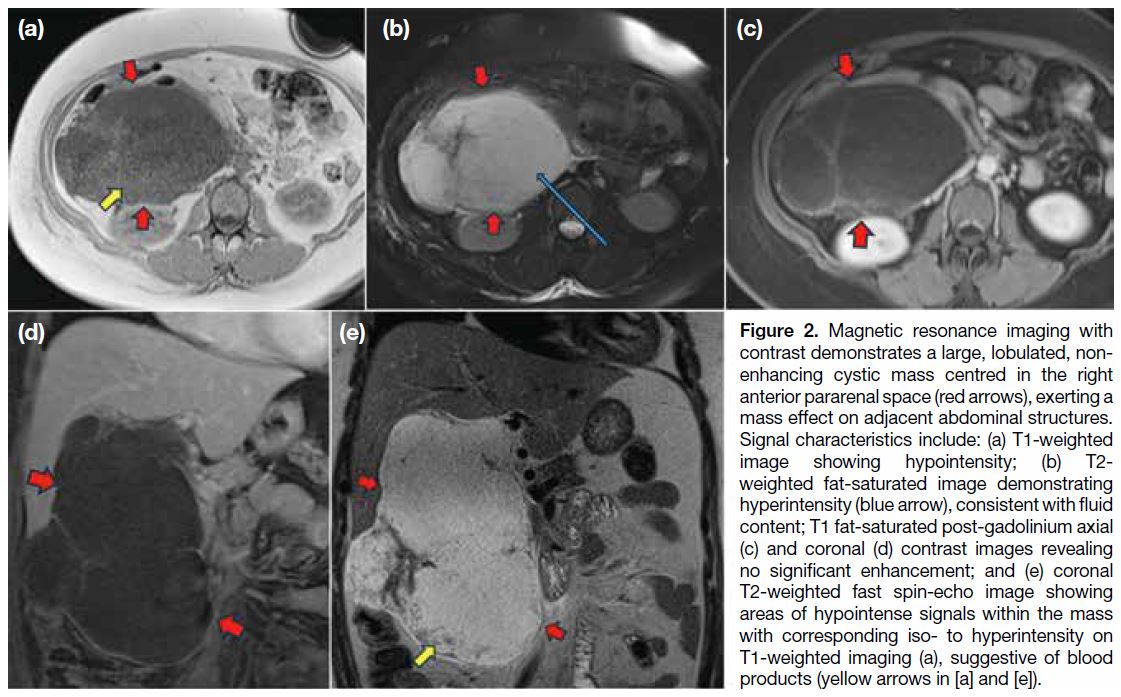
Subsequent magnetic resonance cholangiopancreatography
with contrast (Figure 2) of the patient showed a predominantly T2 hyperintense lobulated mass lesion
measuring 10.5 × 14 × 17 cm3 (anteroposterior × transverse × craniocaudal) in the right anterior
pararenal space, extending superiorly to the subhepatic
region. It impinged on the inferior surface of the liver
and displaced the right mesocolic region inferiorly.
Some T1-weighted iso- to hyperintense signals with
corresponding T2-weighted hypointense signals were also seen, suggestive of blood products. There was
no diffusion restriction abnormality nor significant
enhancement. Compression of the adjacent duodenum,
pancreatic head/uncinate process, inferior vena cava,
right kidney and renal vessels was evident. The
pancreatic duct was not dilated. The main portal vein,
common hepatic artery and common bile ducts were
partially encased but did not appear infiltrated by the mass. There were no enlarged lymph nodes. Mild
abdominopelvic ascites was present.
Figure 2. Magnetic resonance imaging with
contrast demonstrates a large, lobulated, non-enhancing
cystic mass centred in the right
anterior pararenal space (red arrows), exerting a
mass effect on adjacent abdominal structures.
Signal characteristics include: (a) T1-weighted
image showing hypointensity; (b) T2-weighted fat-saturated image demonstrating
hyperintensity (blue arrow), consistent with fluid
content; T1 fat-saturated post-gadolinium axial
(c) and coronal (d) contrast images revealing
no significant enhancement; and (e) coronal
T2-weighted fast spin-echo image showing
areas of hypointense signals within the mass
with corresponding iso- to hyperintensity on
T1-weighted imaging (a), suggestive of blood
products (yellow arrows in [a] and [e]).
Laboratory examinations showed elevated level of
carbohydrate antigen 19-9 at 269.40 U/mL (normal
value = 0.00-37.00). Carcinoembryonic antigen level
was normal at 1.12 ng/mL (normal value < 2.5).
Initial considerations were retroperitoneal lymphatic
malformation or mucinous cystic neoplasm of the
pancreas.
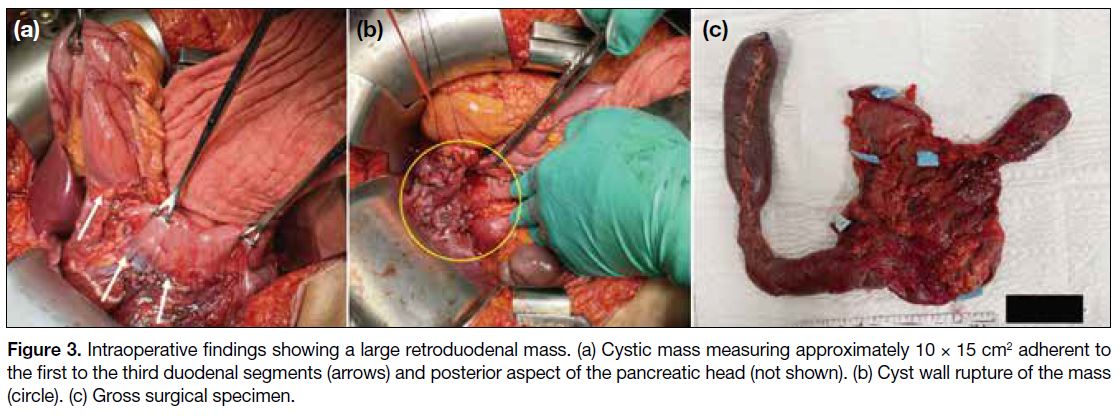
The patient underwent exploratory laparotomy (Figure 3)
that revealed a cystic mass adherent to the first to the third
duodenal segments and posterior aspect of the pancreatic head. It measured about 10 × 15 cm2 (width × length) and
displaced the portal vein and superior mesenteric vein
anteromedially, the duodenum, pancreatic head, vena
cava, and the right kidney posteriorly. The cystic mass
insinuated in between the portal vein and common bile
duct.
Figure 3. Intraoperative findings showing a large retroduodenal mass. (a) Cystic mass measuring approximately 10 × 15 cm2 adherent to
the first to the third duodenal segments (arrows) and posterior aspect of the pancreatic head (not shown). (b) Cyst wall rupture of the mass
(circle). (c) Gross surgical specimen.
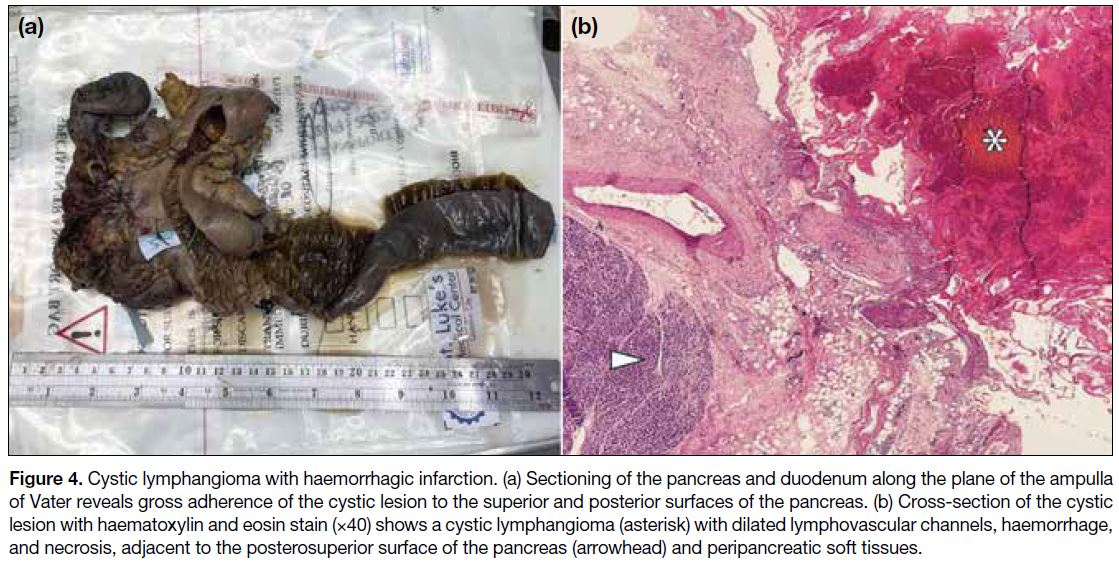
Surgical rupture of the cyst drained approximately 1 L of dark serosanguineous fluid. Pancreaticoduodenectomy
was then performed. Histopathology revealed a
pancreatic cystic lymphangioma with haemorrhagic
infarction (Figure 4). There was no evidence of dysplasia
or malignancy. The patient’s postoperative course was
unremarkable.
Figure 4. Cystic lymphangioma with haemorrhagic infarction. (a) Sectioning of the pancreas and duodenum along the plane of the ampulla
of Vater reveals gross adherence of the cystic lesion to the superior and posterior surfaces of the pancreas. (b) Cross-section of the cystic
lesion with haematoxylin and eosin stain (×40) shows a cystic lymphangioma (asterisk) with dilated lymphovascular channels, haemorrhage,
and necrosis, adjacent to the posterosuperior surface of the pancreas (arrowhead) and peripancreatic soft tissues.
DISCUSSION
Lymphangiomas are benign lymphatic malformations in
the pancreas as a result of blockage of lymphatic outflow.
Most cases are asymptomatic and an incidental finding
on imaging. Nonetheless, the patient may present with
symptoms secondary to mass effect such as abdominal
pain, discomfort, and a palpable abdominal mass.[3] [4] There
is a female preponderance with mean age at presentation
of 28.9 years.[5] Laboratory tests including tumour markers
are usually normal.[5] In our case, there were signs and
symptoms of acute abdomen and an elevated level of
carbohydrate antigen 19-9, characteristics that may be
atypical for pancreatic lymphangioma.
Radiological imaging has a role in preoperative
assessment of pancreatic lymphangiomas. These lesions
may mimic other pancreatic cystic masses including
mucinous cystic neoplasm, pseudocysts and cystic
neuroendocrine tumours, making preoperative diagnosis
difficult.[6] [7] On CT and MRI, pancreatic lymphangiomas
appear as encapsulated homogeneous cystic masses,
as in our patient. These lesions may show enhancing
septations and occasionally contain phleboliths.[7] The
presence of T1 hyperintense signals within cystic masses
indicates lipid, highly proteinaceous, or haemorrhagic
content.[8] In our case, it is interesting to note that the
suggestive blood products present within the mass may
have been congruent with the haemorrhagic infarction
seen intraoperatively. To the best of our knowledge,
there have been no prior reported cases of haemorrhagic
lymphangiomas arising in the pancreas.
On histopathology, lymphangiomas appear as multilocular cysts representing dilated lymphatic
channels containing serous, serosanguineous, or chylous
fluid. The cyst walls are lined with endothelial cells
and are composed of varying degrees of collagenous
connective tissue and smooth muscle.[7]
Endoscopic ultrasound-guided fine needle aspiration
may help establish a definitive preoperative diagnosis
and enable conservative management of asymptomatic
patients.[9] Future studies are encouraged to establish
guidelines for management of pancreatic lymphatic
malformations.[9]
The treatment of choice of pancreatic lymphangiomas is
complete surgical resection accompanied by a low chance
of recurrence. Overall prognosis is excellent. Surgery
is often required for symptom control or diagnosis.[10]
An imaging study on follow-up is an alternative for asymptomatic patients.
CONCLUSION
Although rare, pancreatic lymphangiomas should
be included in the differential diagnoses of complex
pancreatic cystic lesions, especially in asymptomatic
women. Preoperative diagnosis with diagnostic imaging
is usually inconclusive but may determine the extent
of the mass and its relationship to adjacent structures.
A combination of imaging studies and analysis on
endoscopic ultrasound-guided fine needle aspiration can
provide a definitive preoperative diagnosis.
Since lymphangiomas are considered benign,
a conservative approach may be reasonable in
asymptomatic patients and stable lesions once tissue
diagnosis is confirmed. Nonetheless these lesions may be
locally invasive. Radiological imaging, particularly CT
and MRI, may play a role in determining complications
such as obstruction, rupture, or infection that warrant
surgical intervention. Guidelines for appropriate
selection of conservative approach versus surgical
intervention should be established.
REFERENCES
1. Carvalho D, Costa M, Russo P, Simas L, Baptista T, Ramos G.
Cystic pancreatic lymphangioma—diagnostic role of endoscopic
ultrasound. GE Port J Gastroenterol. 2016;23:254-8. Crossref
2. Karajgikar J, Deshmukh S. Pancreatic lymphangioma: a case report
and literature review. J Comput Assist Tomogr. 2019;43:242-4. Crossref
3. Viscosi F, Fleres F, Mazzeo C, Vulcano I, Cucinotta E. Cystic
lymphangioma of the pancreas: a hard diagnostic challenge between
pancreatic cystic lesions—review of recent literature. Gland Surg.
2018;7:487-92. Crossref
4. Chen D, Feng X, Lv Z, Xu X, Ding C, Wu J. Cystic lymphangioma
of pancreas: a rare case report and review of the literature. Medicine
(Baltimore). 2018;97:e11238. Crossref
5. Leung TK, Lee CM, Shen LK, Chen YY. Differential diagnosis of
cystic lymphangioma of the pancreas based on imaging features. J
Formos Med Assoc. 2006;105:512-7. Crossref
6. Anbardar MH, Soleimani N, Aminzadeh Vahedi A, Malek-Hosseini
SA. Large cystic lymphangioma of pancreas mimicking mucinous
neoplasm: case report with a review of histological differential
diagnosis. Int Med Case Rep J. 2019;12:297-301. Crossref
7. Manning MA, Srivastava A, Paal EE, Gould CF, Mortele KJ.
Nonepithelial neoplasms of the pancreas: radiologic-pathologic
correlation, part 1—benign tumors: from the radiologic pathology
archives. Radiographics. 2016;36:123-41. Crossref
8. Stoupis C, Ros PR, Williams JL. Hemorrhagic lymphangioma
mimicking hemoperitoneum: MR imaging diagnosis. J Magn Reson
Imaging. 1993;3:541-2. Crossref
9. Chen Q, Wang Y, Wang J, Hou W, Zou B, Cheng B. Diagnosis
of pancreatic cystic lymphangioma in an 11-year-old boy with
endoscopic ultrasound-guided fine needle aspiration: a case report.
Int J Hepatobiliary Pancreat Dis. 2016;6:114-20. Crossref
10. Cherk M, Nikfarjam M, Christophi C. Retroperitoneal
lymphangioma. Asian J Surg. 2006;29:51-4. Crossref