Giant Cell Tumour of Bone as the Cause of Vertebra Plana in Young Patients: Two Case Reports
CASE REPORT
Hong Kong J Radiol 2025 Mar;28(1):e44-49 | Epub 19 March 2025
Giant Cell Tumour of Bone as the Cause of Vertebra Plana in Young Patients: Two Case Reports
LY Lam, TWY Chin, KC Lai, MK Chan
Department of Diagnostic and Interventional Radiology, Queen Elizabeth Hospital, Hong Kong SAR, China
Correspondence: Dr Y Lam, Department of Diagnostic and Interventional Radiology, Queen Elizabeth Hospital, Hong Kong SAR, China. Email: lly858@ha.org.hk
Submitted: 29 August 2023; Accepted: 21 March 2024.
Contributors: All authors designed the study. LYL acquired the data. All authors analysed the data. LYL drafted the manuscript. All authors critically revised the manuscript for important intellectual content. All authors had full access to the data, contributed to the study, approved the final version for publication, and take responsibility for its accuracy and integrity.
Conflicts of Interest: All authors have disclosed no conflicts of interest.
Funding/Support: This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data Availability: All data generated or analysed during the present study are available from the corresponding author on reasonable request.
Ethics Approval: The patients were treated in accordance with the Declaration of Helsinki. Informed patient consent was obtained for Case 1 for the publication of the case report. Case 2 was lost to follow-up and its status was disclosed to the Kowloon Central Cluster and Kowloon East Cluster Research Ethics Committee of Hospital Authority, Hong Kong when approval was sought (Ref No.: IRB-2023-313). Potential identifiers
have been removed to minimise the risk of identification.
INTRODUCTION
Vertebra plana is a form of complete compression
fracture of the vertebral body. Although Langerhans
cell histiocytosis (LCH) is the most frequent associated
entity,[1] there are other differential diagnoses such as
giant cell tumour (GCT) of bone. We describe two cases
of vertebra plana in young patients with GCT as the
underlying cause.
CASE PRESENTATIONS
Case 1
A 30-year-old female presented to our orthopaedics
department in September 2022 with back pain. She
reported no history of trauma or constitutional symptoms.
Physical examination revealed kyphosis at the upper
thoracic spine and no focal neurological deficits. Initial
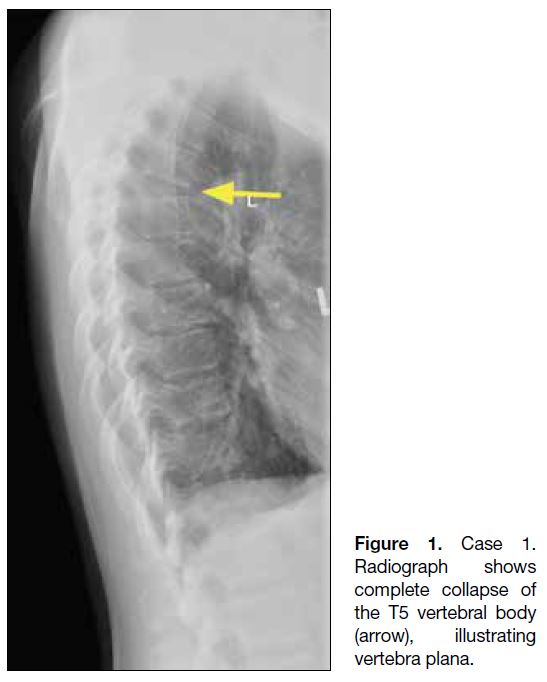
radiograph demonstrated vertebra plana of the T5 vertebra
with an ill-defined left pedicle (Figure 1). Positron
emission tomography–computed tomography (PET/CT)
showed a pathological compression fracture of the T5
vertebra (maximum standardised uptake value [SUVmax]
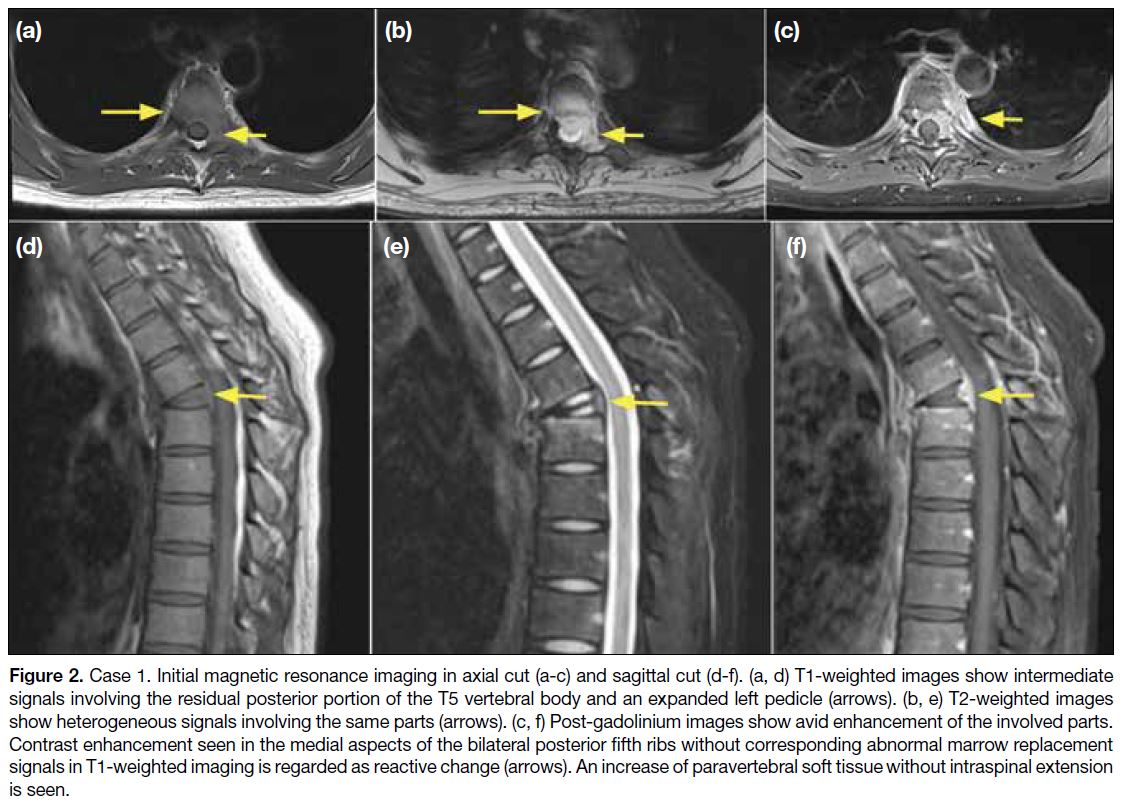
= 4.6) and no lung masses or other hypermetabolic foci. Magnetic resonance imaging (MRI) revealed
complete collapse of the T5 vertebra with involvement
of the left transverse process, lamina and pedicle. The
residual posterior portion of the T5 vertebra and an
expanded left pedicle showed T1-weighted intermediate
and T2-weighted heterogeneous signals with contrast
enhancement (Figure 2). There was increased paravertebral soft tissue without intraspinal extension.
Figure 1. Case 1.
Radiograph shows complete collapse of the T5 vertebral body (arrow), illustrating vertebra plana.
Figure 2. Case 1. Initial magnetic resonance imaging in axial cut (a-c) and sagittal cut (d-f). (a, d) T1-weighted images show intermediate
signals involving the residual posterior portion of the T5 vertebral body and an expanded left pedicle (arrows). (b, e) T2-weighted images
show heterogeneous signals involving the same parts (arrows). (c, f) Post-gadolinium images show avid enhancement of the involved parts.
Contrast enhancement seen in the medial aspects of the bilateral posterior fifth ribs without corresponding abnormal marrow replacement
signals in T1-weighted imaging is regarded as reactive change (arrows). An increase of paravertebral soft tissue without intraspinal extension
is seen.
CT-guided biopsy of the T5 vertebra was performed
in September 2022. Pathology revealed a giant cell-rich
lesion with H3F3A G34W mutation, suggestive
of GCT of bone. The patient had been treated with
denosumab since November 2022. After the third
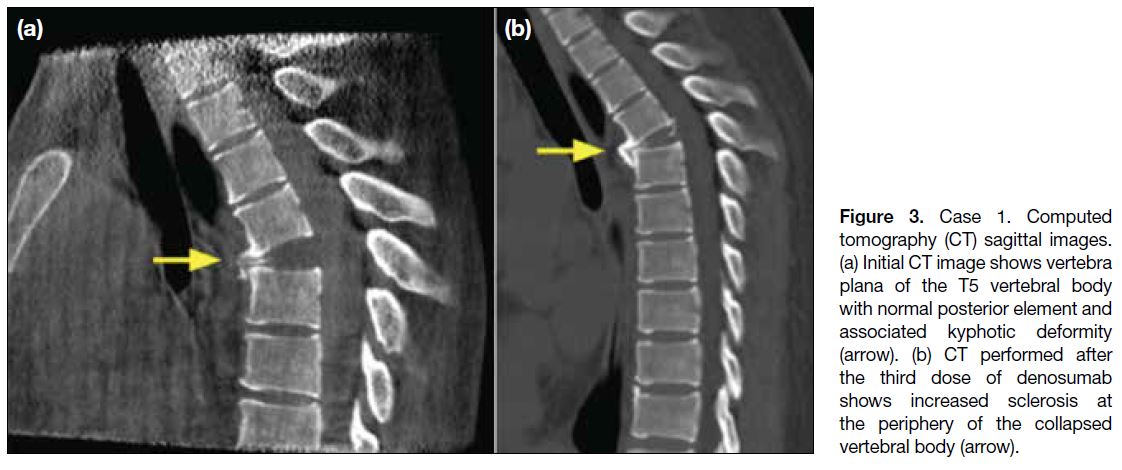
dose of denosumab, follow-up CT showed increased
sclerosis at the periphery and within the tumour
(Figure 3), in keeping with denosumab effect. Follow-up
MRI showed that the tumoural involvement was
static. After nine doses of denosumab, total en bloc
spondylectomy of the T5 vertebra was performed in
May 2023. Final pathology of the T5 vertebra confirmed
the diagnosis of GCT of bone.
Figure 3. Case 1. Computed
tomography (CT) sagittal images.
(a) Initial CT image shows vertebra
plana of the T5 vertebral body
with normal posterior element and
associated kyphotic deformity
(arrow). (b) CT performed after
the third dose of denosumab
shows increased sclerosis at
the periphery of the collapsed
vertebral body (arrow).
Case 2
A male adolescent was referred to a local hospital in
March 2019. He presented with a 1-month history of
neck pain radiating to the arms, with no history of trauma
or constitutional symptoms. Physical examination
revealed reduced range of movement due to pain and
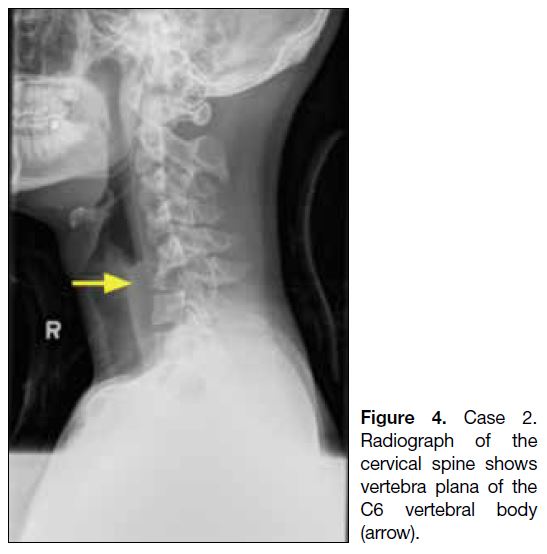
no neurological deficits. Radiograph showed vertebra
plana of the C6 vertebra (Figure 4). PET showed
complete collapse of the C6 vertebral body with a rim
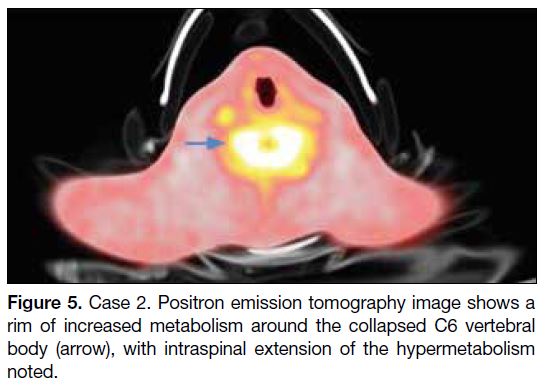
of hypermetabolism (SUVmax = 9.7), with involvement
of the left lamina (SUVmax = 7.4) and intraspinal
extension (Figure 5). There were no lung masses or other
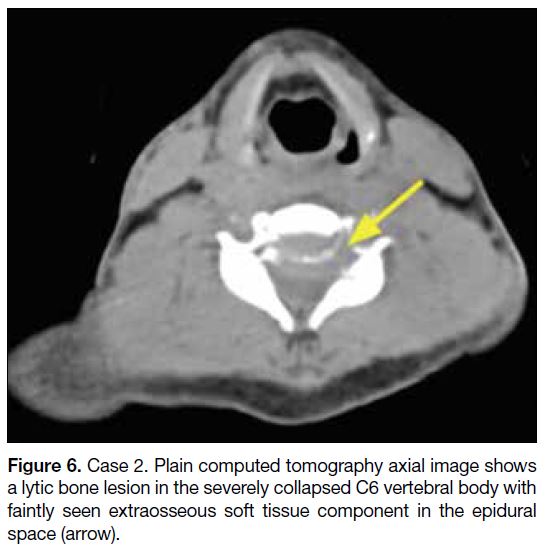
hypermetabolic foci. CT revealed vertebra plana of the
C6 vertebral body with an underlying lytic bone lesion
involving bilateral pedicles and partes interarticulares
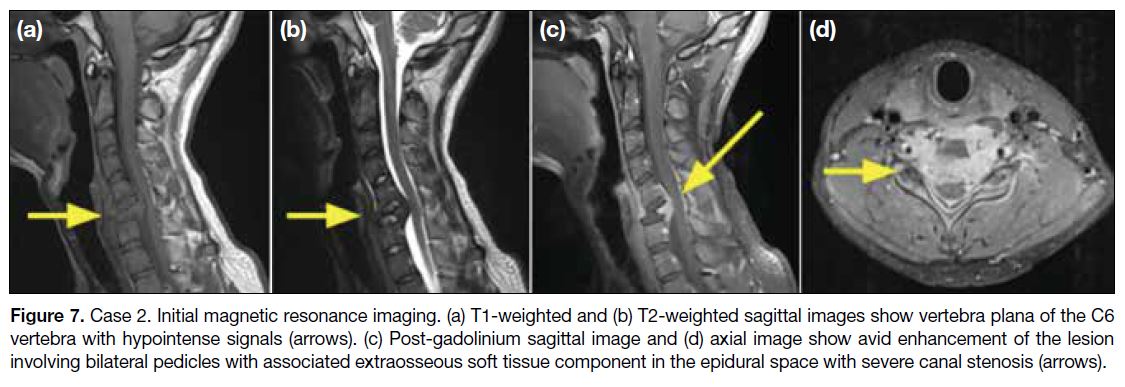
(Figure 6). MRI showed the lesion with T1- and T2-weighted hypointensity, avid contrast enhancement, an
extraosseous epidural soft tissue component and severe
canal stenosis (Figure 7).
Figure 4. Case 2.
Radiograph of the cervical spine shows vertebra plana of the C6 vertebral body (arrow).
Figure 5. Case 2. Positron emission tomography image shows a
rim of increased metabolism around the collapsed C6 vertebral
body (arrow), with intraspinal extension of the hypermetabolism
noted.
Figure 6. Case 2. Plain computed tomography axial image shows
a lytic bone lesion in the severely collapsed C6 vertebral body with
faintly seen extraosseous soft tissue component in the epidural
space (arrow).
Figure 7. Case 2. Initial magnetic resonance imaging. (a) T1-weighted and (b) T2-weighted sagittal images show vertebra plana of the C6
vertebra with hypointense signals (arrows). (c) Post-gadolinium sagittal image and (d) axial image show avid enhancement of the lesion
involving bilateral pedicles with associated extraosseous soft tissue component in the epidural space with severe canal stenosis (arrows).
Pathology from the CT-guided biopsy of the C6 vertebra
revealed a giant cell-rich lesion that showed strong nuclear staining with H3.3 G34W mutant protein,
suggestive of GCT of bone. The patient had been treated
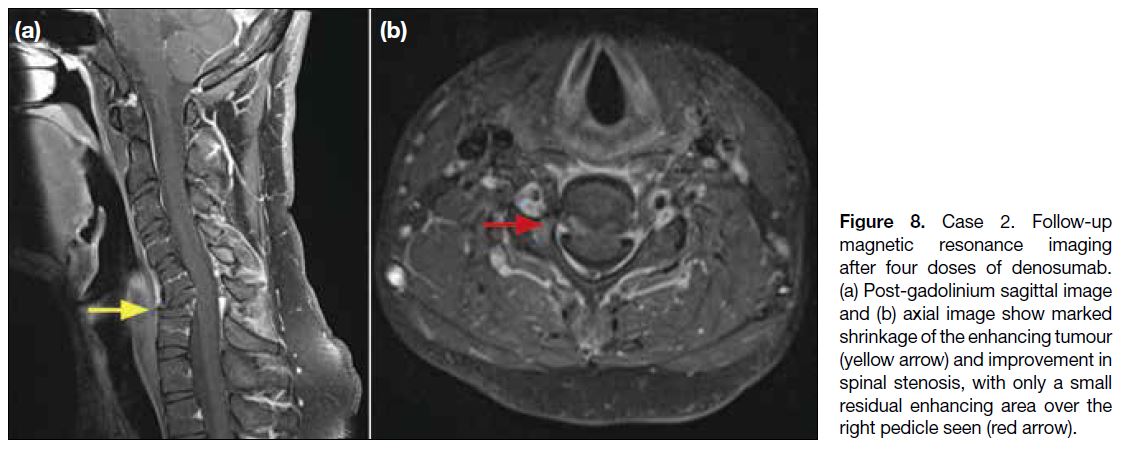
with denosumab since June 2019. After the fourth dose
of denosumab, follow-up MRI showed marked shrinkage
of the tumour with only a small residual enhancing area
over the right pedicle (Figure 8). In view of the good
response to denosumab, the patient and his family opted
for non-operative treatment and continued denosumab
therapy. The patient was lost to follow-up after January
2020.
Figure 8. Case 2. Follow-up
magnetic resonance imaging
after four doses of denosumab.
(a) Post-gadolinium sagittal image
and (b) axial image show marked
shrinkage of the enhancing tumour
(yellow arrow) and improvement in
spinal stenosis, with only a small
residual enhancing area over the
right pedicle seen (red arrow).
DISCUSSION
GCTs are benign bone tumours that can be locally
aggressive and metastasise to the lungs.[2] It is one of the
most common bone tumours and accounts for 4% to
9.5% of primary bone tumours and 18% to 23% of benign
bone tumours.[2] Most patients with GCTs are in their
third or fourth decade of life, with a slight predominance
in females.[3] Our first case of a female in her third decade
exemplifies a typical demographic profile of GCT.
The typical location of GCTs is the ends of long bones,
particularly the distal femur and proximal tibia (up to
65%).[4] The spine is a relatively rare location for GCTs,
accounting for only 7% of all cases.[5] Within the spine,
GCTs most commonly occur at the sacrum, followed by
the thoracic, cervical and lumbar spine in descending
order of frequency.[6]
A large case series[6] showed that 85% of GCTs affecting the spine arose from the vertebral body with involvement
of the vertebral arch, while others arose from the
posterior elements. All cases in the series affected the
vertebral bodies and pedicles.[6] This feature enables us
to differentiate GCTs from other primary spinal bone
tumours such as osteoid osteoma, osteoblastoma and
aneurysmal bone cysts (ABCs), since these tumours
preferentially affect the posterior elements.
Imaging features of GCTs on radiographs include an
expansile appearance and osteolysis. On CT, spinal GCTs
appear as well-defined soft tissue density masses with
a sclerotic rim and without internal mineralised matrix.
They typically show avid contrast enhancement due to
their hypervascular nature. On MRI, spinal GCTs exhibit
low to intermediate T1-weighted signal intensities with
gadolinium enhancement. On T2-weighted images, most
spinal GCTs show heterogeneous high signals with areas
of low signal intensities due to hemosiderin or fibrosis.[7]
Differential diagnoses of a spinal bone lesion showing
T2-weighted hypointense signals are limited and include
myelofibrosis, osteoblastic metastases and spinal GCT.[7]
The first two do not match our cases. In myelofibrosis,
the diseased bone shows homogeneous low signals, and
myelofibrosis does not cause vertebra plana. Our patients’
young age and lack of known primary malignancy do not
suggest metastases.
A spinal GCT may result in collapse of the vertebral
body, ranging from mild collapse to rarely, vertebra
plana, as seen in our cases. Vertebra plana was first
described by Calve in 1925 as ‘extreme collapse of a
vertebral body with sparing of the posterior elements
and slightly widened adjacent disc spaces’.[8] Later, most
reports described it as collapse of a single vertebral body
with increased density, normal adjacent intervertebral
discs and increased height of intervertebral space by at
least one-third of normal.
Vertebra plana is often thought to be pathognomonic for
LCH in young patients. This is a false assumption since
many different conditions can cause vertebra plana,
including ABCs, GCTs, lymphomas, and metastases.[9]
As LCH is more commonly seen in paediatric patients,[5] [10]
it is an unlikely diagnosis based on our patients’
demographics. In addition, LCH causes prominent
oedema in the adjacent bone and soft tissues, which are
absent in our cases. Spinal lymphoma can present as an
osteolytic lesion and cause vertebra plana, though this is
exceedingly rare and usually involves multiple levels.[11]
Our cases also do not present with any B symptoms.
An ABC typically affects the posterior elements with
pure cystic components, making it unlikely in our cases.
The age of our patients is not typical for plasmacytoma.
Infection is also an important differential diagnosis but
the intact endplates and intervertebral discs, absence of
fluid collection, and the chronicity in our cases speak
against infection.
The ideal modality of treatment for spinal GCTs is
complete surgical resection. Some cases may present
technical difficulties, especially if the tumour is located
in the cervical spine with complicated surrounding
anatomy, as seen in Case 2.
Denosumab has been suggested as an alternative
therapy. It is a human monoclonal antibody that binds
the receptor activator of nuclear factor kappa-beta ligand,
preventing activation of its receptor on the surface
of giant cells and osteoclasts. This inhibits osteoclast formation and reduces bone resorption in GCTs. A
study reported that 96% of patients with surgically
unsalvageable GCTs treated with denosumab showed no
progression.[12] A report described complete regression of
spinal GCTs following denosumab treatment without the
need for surgical resection.[13]
Additionally, denosumab acts as a preoperative adjunct
in cases of spinal or pelvic GCT. It reduces extraosseous
soft tissue lesions, hardens the GCT, and therefore
facilitates en bloc resection, as seen in Case 1. For GCTs
in the extremities, denosumab treatment is not routinely
recommended as curettage remains the mainstay of
treatment and denosumab prior to curettage is known to
increase the local recurrence rate,[14] possibly due to the
effect of increased osteosclerosis, rendering it difficult to
identify the tumoural involvement intraoperatively.
The optimal duration of denosumab treatment is
unknown. Some authors recommend a neoadjuvant
therapy duration of 3 to 4 months[15] but further studies
are required to determine the optimal duration.
In summary, we report two cases of vertebra plana in
young patients caused by GCT of bone. Although
GCT remains a rare cause of vertebra plana, it should
be considered as a differential diagnosis. Surgery is the
mainstay treatment while denosumab therapy is helpful
as a preoperative adjunct or as an alternative treatment in
surgically challenging cases.
REFERENCES
1. Papagelopoulos PJ, Currier BL, Galanis E, Grubb MJ, Pritchard DJ,
Ebersold MJ. Vertebra plana caused by primary Ewing sarcoma:
case report and review of the literature. J Spinal Disord Tech.
2002;15:252-7. Crossref
2. Murphey MD, Nomikos GC, Flemming DJ, Gannon FH,
Temple HT, Kransdorf MJ. From the archives of AFIP. Imaging
of giant cell tumor and giant cell reparative granuloma of bone:
radiologic-pathologic correlation. Radiographics. 2001;21:1283-309. Crossref
3. Kafchitsas K, Habermann B, Proschek D, Kurth A, Eberhardt C.
Functional results after giant cell tumor operation near knee
joint and the cement radiolucent zone as indicator of recurrence.
Anticancer Res. 2010;30:3795-9.
4. Chakarun CJ, Forrester DM, Gottsegen CJ, Patel DB, White EA,
Matcuk GR Jr. Giant cell tumor of bone: review, mimics, and new
developments in treatment. Radiographics. 2013;33:197-211. Crossref
5. Zulkharnain EM, Muhamad Ariffin MH, Tan JA, Afiq Zakaria MA. Giant cell tumour of the spine. Cureus. 2024;16:e71166. Crossref
6. Shi LS, Li YQ, Wu WJ, Zhang ZK, Gao F, Latif M. Imaging
appearance of giant cell tumour of the spine above the sacrum. Br
J Radiol. 2015;88:20140566. Crossref
7. Badkhane S, Singh S, Dixit R, Garg A. T2 dark lesions of the
musculoskeletal system: a pictorial essay. Hong Kong J Radiol. 2021;24:e15-23. Crossref
8. Calvé J. A localized affection of the spine suggesting osteochondritis of the vertebral body, with the clinical aspect of Pott’s disease. J Bone Joint Surg. 1925;7:41-6.
9. Baghaie M, Gillet P, Dondelinger RF, Flandroy P. Vertebra plana:
benign or malignant lesion? Pediatr Radiol. 1996;26:431-3. Crossref
10. Jezierska M, Stefanowicz J, Romanowicz G, Kosiak W, Lange M.
Langerhans cell histiocytosis in children—a disease with many
faces. Recent advances in pathogenesis, diagnostic examinations
and treatment. Postepy Dermatol Alergol. 2018;35:6-17. Crossref
11. Zahid M, Ahamed S, Jain JK, Chabra R. Vertebra plana with
paraplegia in a middle-aged woman caused by B-cell lymphoma:
a case report. Case Rep Orthop. 2012;2012:101506. Crossref
12. Chawla S, Henshaw R, Seeger L, Choy E, Blay JY, Ferrari S,
et al. Safety and efficacy of denosumab for adults and skeletally mature adolescents with giant cell tumour of bone: interim analysis
of an open-label, parallel-group, phase 2 study. Lancet Oncol.
2013;14:901-8. Crossref
13. Mattei TA, Ramos E, Rehman AA, Shaw A, Patel SR, Mendel E.
Sustained long-term complete regression of a giant cell tumor of
the spine after treatment with denosumab. Spine J. 2014;14:e15-21. Crossref
14. Tsukamoto S, Mavrogenis AF, Kido A, Errani C. Current concepts
in the treatment of giant cell tumors of bone. Cancers (Basel).
2021;13:3647. Crossref
15. Borkowska AM, Szumera-Ciećkiewicz A, Szostakowski B,
Pieńkowski A, Rutkowski PL. Denosumab in giant cell tumor
of bone: multidisciplinary medical management based on
pathophysiological mechanisms and real-world evidence. Cancers
(Basel). 2022;14:2290. Crossref