Artificial Intelligence for Contouring and Treatment Planning in Locoregional Radiotherapy Including Internal Mammary Nodal Irradiation for Breast Cancer
ORIGINAL ARTICLE
Hong Kong J Radiol Radiol 2025 Mar;28(1):e23-34 | Epub 24 March 2025
Artificial Intelligence for Contouring and Treatment Planning in Locoregional Radiotherapy Including Internal Mammary Nodal Irradiation for Breast Cancer
CCC Wong, YL Wong, KF Choi, SCY Ng, ASY Chan, MY Luk, KK Yuen
Department of Clinical Oncology, Queen Mary Hospital, Hong Kong SAR, China
Correspondence: Dr CCC Wong, Department of Clinical Oncology, Queen Mary Hospital, Hong Kong SAR, China. Email: wcc742@ha.org.hk
Submitted: 10 July 2024; Accepted: 14 November 2024.
Contributors: All authors designed the study. CCCW, YLW, KFC, SCYN, ASYC and MYL acquired the data. CCCW and YLW analysed the
data. CCCW drafted the manuscript and critically revised the manuscript for important intellectual content. All authors had full access to the
data, contributed to the study, approved the final version for publication, and take responsibility for its accuracy and integrity.
Conflicts of Interest: As an editor of the journal, CCCW was not involved in the peer review process. Other authors have disclosed no conflicts of interest.
Funding/Support: This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data Availability: All data generated or analysed during the present study are available from the corresponding author on reasonable request.
Ethics Approval: This research was approved by the Institutional Review Board of The University of Hong Kong/Hospital Authority Hong Kong West Cluster, Hong Kong (Ref No.: UW 24-221). The requirement for informed patient consent was waived by the Committee due to the retrospective nature of the research.
Abstract
Introduction
Postoperative internal mammary node (IMN) irradiation in high-risk early-stage breast cancer is
becoming a standard procedure. We assessed the performance of artificial intelligence (AI) in contouring and
treatment planning in locoregional radiotherapy including IMN for breast cancer.
Methods
We included 27 patients who underwent postoperative locoregional radiotherapy. The study consisted of
two parts. For the first part, AI-generated segmentations of target volumes and organs at risk (OARs) were compared
to manual segmentations using four metrics, namely, volume ratio (VR), the 95th percentile Hausdorff distance (95%
HD), surface Dice similarity coefficient (sDSC), and volume Dice similarity coefficient (vDSC). The time needed
for auto-segmentation was recorded. For the second part, AI-generated volumetric modulated arc therapy (VMAT)
and intensity modulated radiation therapy (IMRT) treatment plans using machine learning (ML) were compared to
manually created plans, focusing on dosimetric parameters and planning times.
Results
AI demonstrated excellent performance in segmenting OARs, with high sDSC and vDSC, low 95% HD
and a VR of 1.01 ± 0.03. However, performance for breast and nodal volumes was less robust. The time for auto-segmentation
averaged 27.39 ± 2.53 seconds. In treatment planning, both ML and manual plans achieved satisfactory
target volumes coverage and OAR constraints, except that IMRT (ML) plans did not meet the dose limit for the
ipsilateral lung. VMAT (ML) planning took less time than manual planning, while IMRT (ML) planning took longer
than manual planning.
Conclusion
AI is proficient in segmenting OARs but is less reliable for target volumes. It produces VMAT plans
comparable to manual plans in a shorter timeframe.
Key Words: Artificial intelligence; Breast neoplasms; Organs at risk; Radiotherapy, intensity-modulated
中文摘要
人工智能在乳癌包含內乳淋巴區的局部放射治療的輪廓勾畫及治療規劃之應用
黃卓卓、黃逸霖、蔡國鋒、吳楚儀、陳心妍、陸美儀、袁國強
引言
在高風險早期乳癌進行術後內乳淋巴區放射治療逐漸成為標準程序。我們評估人工智能在乳癌包含內乳淋巴區的局部放射治療的輪廓勾畫及治療規劃方面之表現。
方法
我們納入27名接受了術後局部放射治療的患者。本研究分為兩部分,在第一部分,我們使用以下四個指標比較人工智能產生及人手制訂的靶區體積及危及器官勾畫:(1)體積比;(2)95% Hausdorff距離;(3)表面Dice相似系數;及(4)體積Dice相似系數。我們並紀錄了經人工智能勾畫所需的時間。至於第二部分,我們比較經機器學習人工智能產生的體積調控弧型放射治療(VMAT)及強度調控放射治療(IMRT)計劃與人手制訂計劃,重點為劑量測定參數及規劃時間。
結果
人工智能在勾畫危及器官方面表現優秀,表面Dice相似系數及體積Dice相似系數高,95%Hausdorff距離小,體積比為1.01 ± 0.03,但在乳房及淋巴結體積的表現較不穩定。經人工智能勾畫的時間平均為27.39 ± 2.53秒。在治療規劃,機器學習及人手制訂計劃均達至滿意的靶區體積覆蓋及危及器官劑量限制,唯機器學習IMRT計劃未能符合同側肺的劑量限制。機器學習VMAT計劃需要的時間較人手制訂計劃短,機器學習IMRT計劃需要的時間則較人手制訂計劃長。
結論
人工智能擅長勾畫危及器官,但在勾畫靶區體積方面則較遜色。它能在較短時間內製作可媲美人手制訂的VMAT計劃。
INTRODUCTION
Breast cancer is the most prevalent cancer affecting
women worldwide. According to the World Health
Organization, in 2022, there were approximately 2.3
million new breast cancer diagnoses made globally, and
670,000 deaths.[1] In Hong Kong, approximately 5500 new
breast cancer cases were reported in 2021, with a crude
incidence rate of 138.1 per 100,000 female population.[2]
The treatment of breast cancer typically involves a
multimodal approach, including surgery, radiotherapy,
and systemic therapies. Adjuvant radiotherapy plays
a crucial role in reducing the risk of cancer recurrence
and improving survival, particularly for patients who
have undergone breast conserving surgery or have been
diagnosed with locally advanced breast cancer.
The Danish Breast Cancer Group Internal Mammary
Node study in 2022[3] showed that elective irradiation of
the internal mammary nodes (IMNs) reduced the risk
of distant recurrence and improved long-term survival
of patients with node-positive early breast cancer,
regardless of breast cancer laterality. However, the irradiation of IMNs has remained a controversial topic,
and guidelines for IMN irradiation vary significantly
across different institutes. One controversial issue is
the potential for long-term cardiotoxicity,[4] as IMN
irradiation tends to result in a higher cardiac dose.[5]
The technical complexities involved in radiotherapy
treatment can be challenging, leading to an increase in
the time and resources required to treat each patient.
Over the past decade, there has been rapid advancement
in artificial intelligence (AI) and deep learning
technology, which have been increasingly utilised in
radiation oncology.[6] The AI-powered tools can perform
segmentation, treatment planning, and delivery,[7] with
the aim of improving the quality and consistency of
the treatment delivered, and enhancing the efficiency
of the work of oncologists and radiation therapists.
Ultimately, the incorporation of AI in radiation oncology
seeks to streamline hospital operations, thus benefiting
a greater number of patients within the demanding
clinical environment. In the past few years, the use of
deep learning and convolutional neural networks on
segmentation as well as treatment planning in breast cancer radiotherapy has been explored. These studies
have shown promising results, with the automated
techniques demonstrating comparable quality to manual
approaches.[8] [9] [10] [11] [12]
In this article, we sought to compare the performance
of AI to that of manual approaches, in the context of
segmentation and treatment planning in radiotherapy of
IMNs in breast cancer.
METHODS
Patient Enrolment
The study included the data from 27 breast cancer
patients, comprising 14 patients with left-sided breast
cancer and 13 with right-sided breast cancer. All patients
had undergone either mastectomy or breast conserving
surgery (including lumpectomy and cryotherapy), as
well as axillary surgery (including sentinel lymph node
biopsy and axillary dissection). Five of the mastectomy
patients also had undergone breast reconstruction. All
27 patients had received postoperative locoregional
radiotherapy including irradiation of clinically evident
IMN (i.e., lymph nodes that are suspected to be involved
by malignancy on imaging) at the dose of 40 Gy in
15 fractions over 3 weeks between 2017 and 2023. A
tumour bed electron boost was administered at a dose
of 10 Gy in five fractions over 1 week to patients who
had undergone breast conserving surgery. Patient
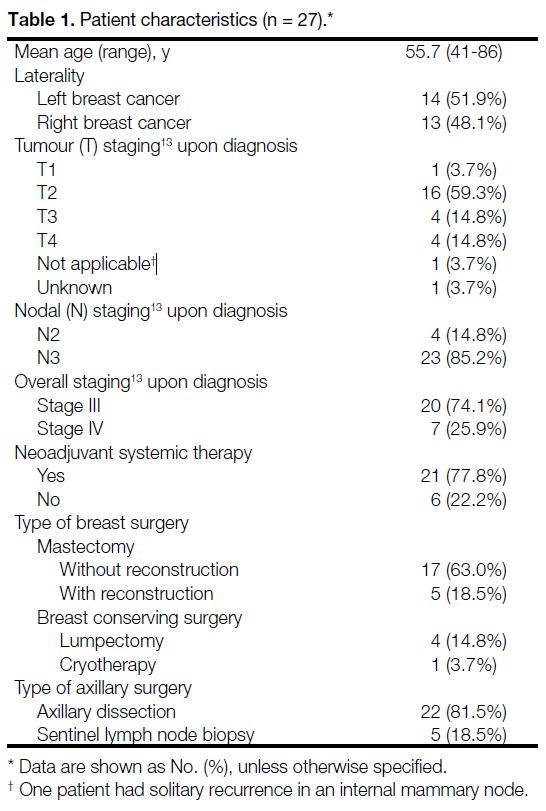
characteristics (including cancer staging[13]) are presented in Table 1.
Table 1. Patient characteristics (n = 27).
Image Acquisition
All patients were positioned in the supine orientation on a breast board (Breastboard MT-350N; CIVCO, Orange
City [FL], US), with the arms abducted and holding onto
a central rod. Simulation was performed using computed
tomography (CT), with a slice separation of 2.5 mm,
covering the region from the jaw to the mid-abdomen.
For patients who had undergone mastectomy without
reconstruction, a 0.5-cm skin bolus was administered to
the chest wall in the planning process.
The CT data were retrieved and exported to treatment
planning software Eclipse[14] (version 15.6; Varian
Medical Systems, Palo Alto [CA], US) and RayStation
12A deep learning segmentation software[15] (RaySearch
Laboratories, Stockholm, Sweden) to perform contouring
and treatment planning.
Contouring
This study comprises two parts. In the first part of the study, two sets of contours were generated from the
patients’ CT data. The performance of auto-segmentation
was assessed by comparing its results to the manual
contouring done by the oncologists.
Manual Contours
Contouring of target volume and organs at risk (OARs)
were done on Eclipse with reference to the European
Society for Radiotherapy and Oncology consensus
guidelines.[16] They were reviewed and approved by two
senior clinical oncologists, each having >10 years of
experience in breast cancer.
Auto-contours or Auto-segmentation
Automatic contouring of target volume and OARs were
created using the deep learning segmentation (RSL
Breast CT_1) planning model within RayStation 12A.
A 5-mm expansion was applied to the breast or chest
wall, axillary levels 3 to 4, and the IMNs from manual
contours to become the planning target volume (PTV).
The PTV was then trimmed by 5 mm underneath the skin, except in slim patients where the thoracic chest
wall was too thin, causing the target volume to disappear
when cropped. The time taken for generation of auto-segmentation
was recorded.
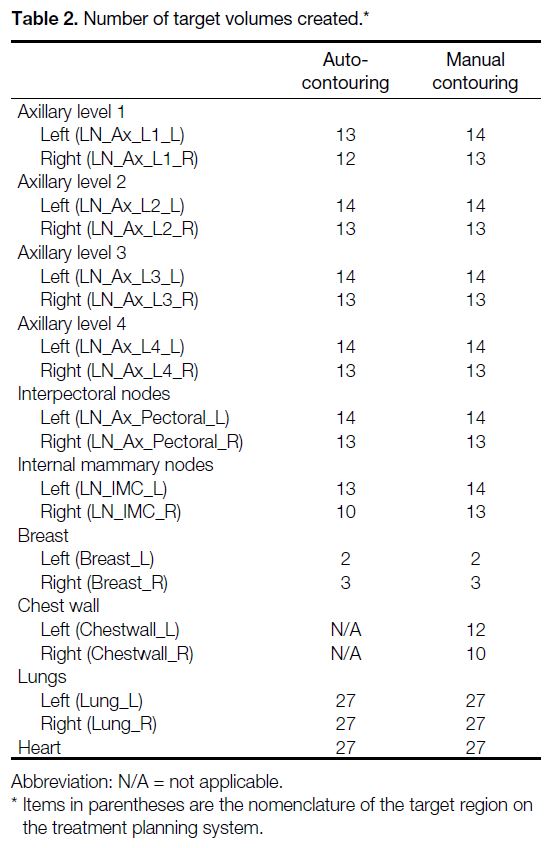
The list and number of target volumes that were created
are presented in Table 2.
Table 2. Number of target volumes created.
Radiotherapy Treatment Planning
In the second part of the study, four virtual treatment
plans were created. These included two plans created
with machine learning (ML): a volumetric modulated
arc therapy ML [VMAT (ML)] plan and an intensity
modulated radiation therapy ML [IMRT (ML)] plan
using a dynamic multileaf collimator with the ML
model RSL-Breast-L-nodes-4005(1.0). Manual VMAT
[VMAT (manual)] and manual IMRT [IMRT (manual)]
plans were generated by the usual manual treatment
planning methods.
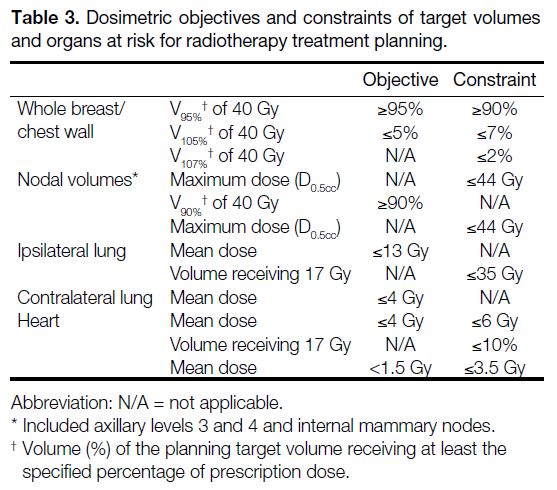
The dosimetric objectives, constraints for target volumes
and OARs used for treatment planning dose optimisation
are presented in Table 3, in accordance with departmental
consensus, The Royal College of Radiologists’ UK
Consensus Statements,[17] and the Danish Breast Cancer
Cooperative Group HYPO II protocol.[18] We recorded
the time needed for automated and manual treatment
planning.
Table 3. Dosimetric objectives and constraints of target volumes
and organs at risk for radiotherapy treatment planning.
Parameters for Comparison
Contouring
The manually contoured and auto-segmented volumes
were compared using the following four geometrical
metrics:
1. Volume ratio (VR)[19]: This metric evaluates the
ratio of the volumes calculated by the test contour
(auto-segmentation) and reference contour (manual
contouring). A VR close to 1 indicates good agreement
between the two contours.
2. The 95th percentile Hausdorff distance (95%
HD)[19] [20]: HD is a surface-based metric of the maximum
perpendicular distance between two sets of points (in
mm). It is used to quantify the differences between the
field edges by manual and auto-contouring. The purpose
of using the 95% HD instead of maximum HD is to
eliminate the impact of small outliers.
3. Surface Dice similarity coefficient (sDSC)[19] [21] [22]:
sDSC is a metric of spatial overlap between two volumes
as a measure of estimated editing. It specifically looks at the overlap between the surfaces of the reference and
test contours, providing an assessment of the contouring
agreement. It has been shown to have a good correlation
with the time required to edit contours.[23] [24]
4. Volume Dice similarity coefficient (vDSC)[19] [22]:
vDSC evaluates the overall volumetric overlap between
the reference and test contours. This gives an indication
of how well the total volumes agree. However, it is not
sensitive to complex boundaries and tends to give higher
scores for larger volumes. It is generally accepted that
volume DSC >0.7 indicates excellent agreement.[25] [26]
Radiotherapy Treatment Planning
Dose-volume histograms (DVHs) were generated in
each treatment plan for review of dosimetric parameters,
including the PTV of the whole breast or chest wall
(V95% [i.e., volume (%) of the PTV receiving at least
95% of the prescription dose] of 40 Gy, V105% of 40 Gy,
V107% of 40 Gy, and maximum dose [D0.5cc]), the PTV of the nodal volumes (V90% of 40 Gy and maximum dose
[D0.5cc]), the mean dose to the ipsilateral lung, the volume
receiving 17 Gy in the ipsilateral lung, the mean dose
to the contralateral lung, the mean dose to the heart, the
volume receiving 17 Gy in the heart, and the mean dose
to the contralateral breast.
Statistical Analyses
Descriptive analyses were stated as mean ± standard deviation; descriptive tables were created for the
comparison of the contouring performance by AI. The
dosimetric parameters for the target volumes and OARs
in different treatment plans were compared using the
Wilcoxon signed rank test for statistical significance. A
p value < 0.05 was considered statistically significant.
RESULTS
Contouring
AI successfully performed auto-segmentation of
parts of the target volumes including axillary level 2,
axillary level 3 (infraclavicular volume), axillary level
4 (supraclavicular volume), interpectoral nodes, breast
(in breast conserving surgery patients), as well as
OARs including the heart and the lungs for all patients.
Nevertheless, it failed in identifying axillary level 1 in
two patients (one on the left side and one on the right
side) and IMNs in four patients (one on the left side
and three on the right side). AI misclassified the region
of the chest wall in mastectomy patients as breast due
to the absence of a specific chest wall segmentation
function on RayStation 12A (Table 4). Thus, the result of
comparison between manual and auto-contouring of the
chest wall was disregarded. Table 2 shows the number
of structures created by auto- and manual contouring,
respectively.
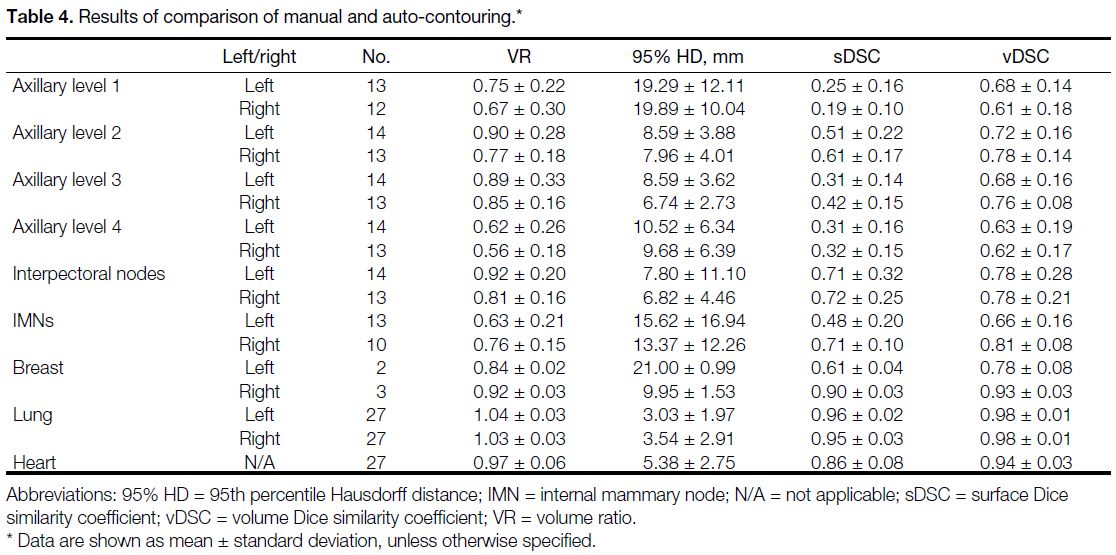
Table 4. Results of comparison of manual and auto-contouring.
Table 4 shows the results of the VR, 95% HD, sDSC,
and vDSC for all structures.
Overall, AI demonstrated excellent performance in
delineating the lungs and heart across all patients. In
contrast, the performance for the target structures, i.e.,
nodal volumes and the breast, was less robust compared
to the OARs, based on the four evaluation metrics used.
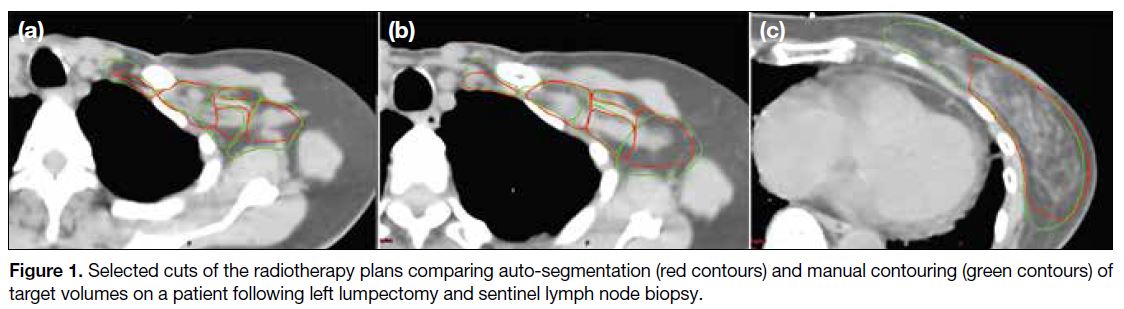
Figure 1 shows the manual contouring and auto-segmentation
of the axillary nodal volume and breast
on selected CT slices. When comparing the contouring
of the nodal volumes, looking at VRs, the AI-generated
structures tended to have smaller volumes compared to
the manual contours, with VRs ranging from 0.56 ± 0.18
to 0.92 ± 0.03. This discrepancy was more pronounced
for contouring of axillary level 4, axillary level 1, and
IMNs (Table 4).
Figure 1. Selected cuts of the radiotherapy plans comparing auto-segmentation (red contours) and manual contouring (green contours) of target volumes on a patient following left lumpectomy and sentinel lymph node biopsy.
The largest 95% HD measurements were observed at
axillary level 1, reaching up to 19.29 ± 12.11 mm for
the left axillary level 1 and 19.89 ± 10.04 mm for the
right axillary level 1, and left breast with the result of
21.00 ± 0.99 mm. This was followed by the IMNs, with
measurements of 15.62 ± 16.94 mm for the left side
and 13.37 ± 12.26 mm for the right side. The 95% HD
measurements for the remaining structures ranged from
6.74 ± 2.73 mm to 10.52 ± 6.34 mm (Table 4).
High sDSC values were observed for interpectoral
nodes, axillary level 2 and the breast. The lowest
agreement was found for axillary level 1. The best results
for vDSC were again observed in the interpectoral nodes,
axillary level 2 and the breast. Conversely, both axillary
level 1 and axillary level 4 had the lowest vDSC values
(Table 4).
Results of nodal volumes in patients who underwent
axillary dissection and sentinel lymph node biopsy
(SLNB) are shown in Tables 5 and 6, respectively. In patients who underwent SLNB instead of axillary
dissection, there was a tendency towards better
performance of auto-segmentation, as indicated by
higher sDSC and vDSC, in comparison to patients who
underwent axillary dissection. However, it is important
to note that a limitation of this analysis is the small
sample size of patients who underwent SLNB.
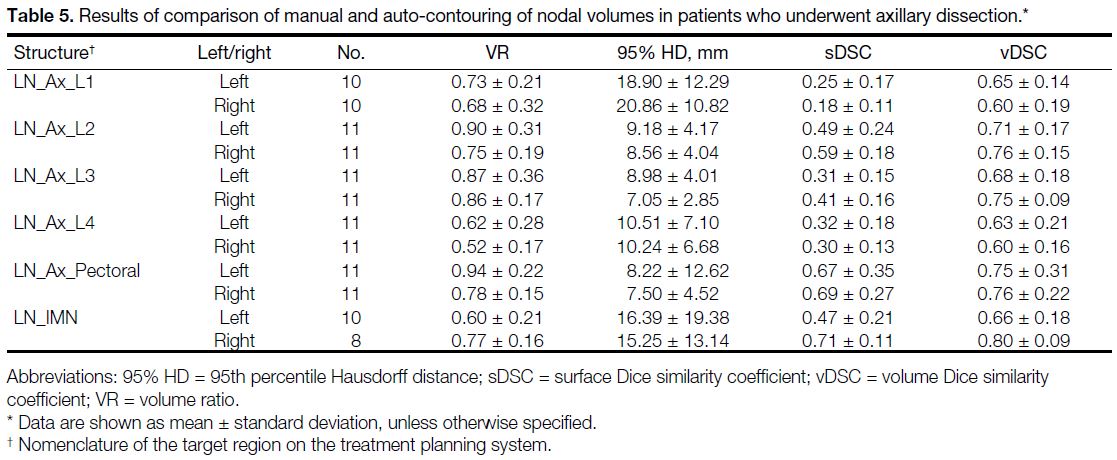
Table 5. Results of comparison of manual and auto-contouring of nodal volumes in patients who underwent axillary dissection.
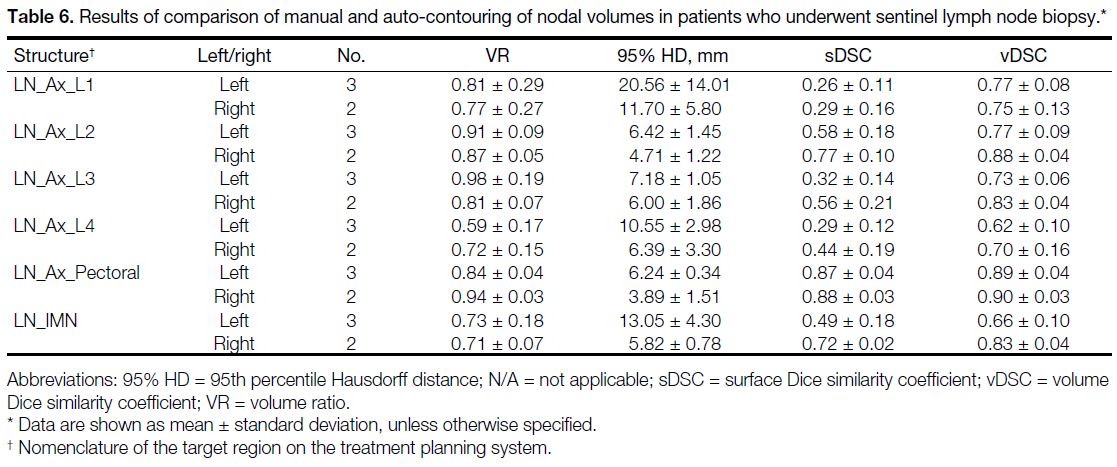
Table 6. Results of comparison of manual and auto-contouring of nodal volumes in patients who underwent sentinel lymph node biopsy.
The time required for auto-segmentation was recorded,
showing an average of 27.39 ± 2.53 seconds for
generation of one contouring.
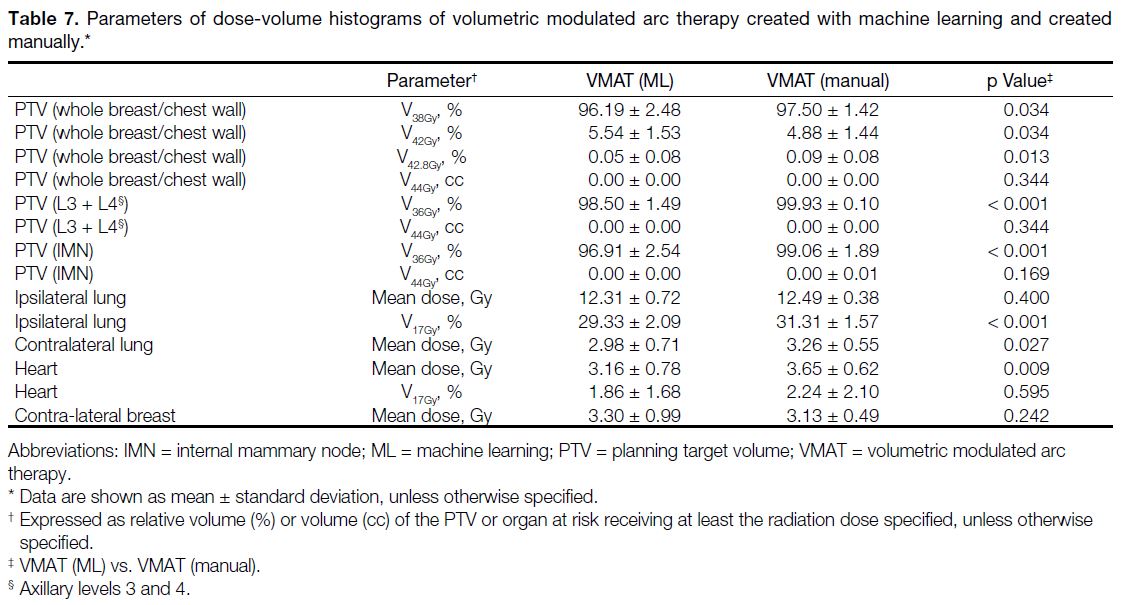
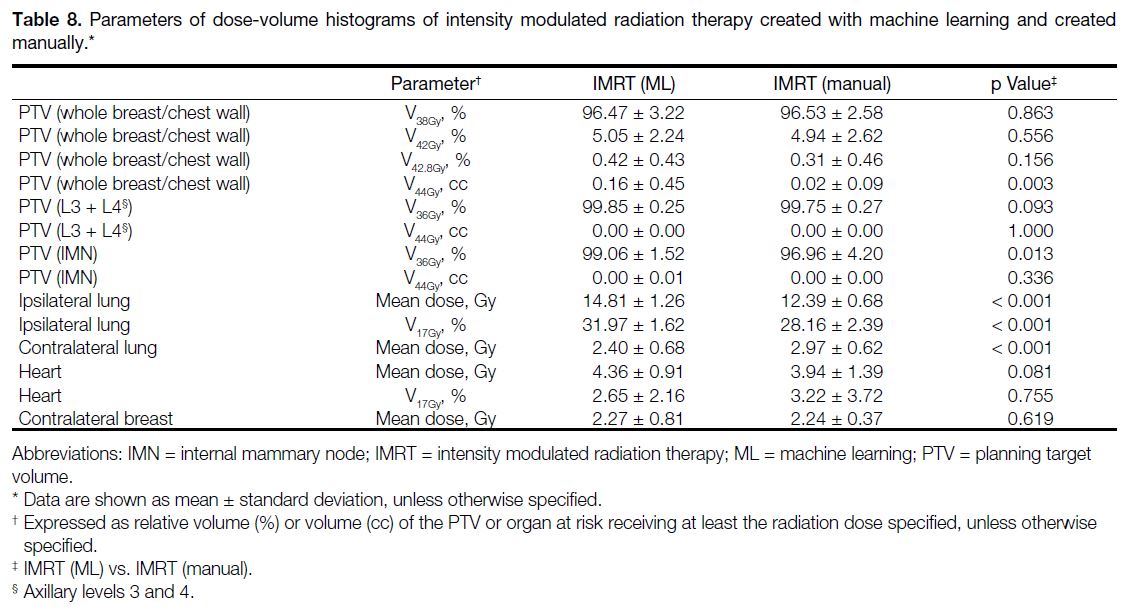
Treatment Planning
DVH parameters of VMAT (ML) and VMAT (manual)
are presented in Table 7, while DVH parameters of
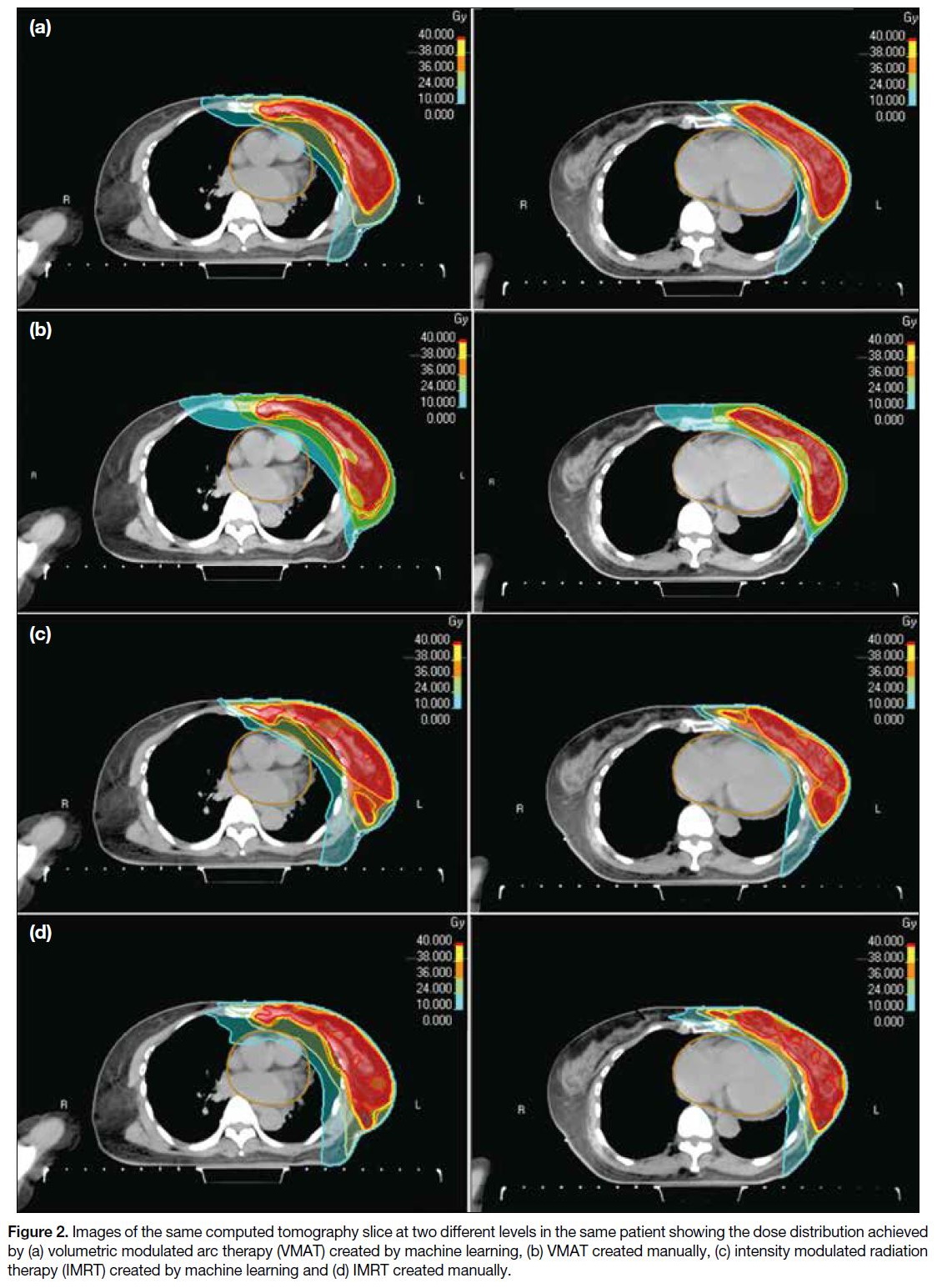
IMRT (ML) and IMRT (manual) are shown in Table 8. Figure 2 illustrates the dose distribution comparisons
between VMAT (ML), VMAT (manual), IMRT (ML)
and IMRT (manual) on selected slices.
Figure 2. Images of the same computed tomography slice at two different levels in the same patient showing the dose distribution achieved
by (a) volumetric modulated arc therapy (VMAT) created by machine learning, (b) VMAT created manually, (c) intensity modulated radiation
therapy (IMRT) created by machine learning and (d) IMRT created manually.
Table 7. Parameters of dose-volume histograms of volumetric modulated arc therapy created with machine learning and created manually.
Table 8. Parameters of dose-volume histograms of intensity modulated radiation therapy created with machine learning and created manually.
All of the four virtual plans achieved satisfactory
performance in covering the target volumes, including
the breast or chest wall and nodal volumes, by fulfilling
the dose constraints. For OARs, IMRT (ML) failed to
achieve the dose limit for mean dose to the ipsilateral
lung, while the other parameters were fulfilled in all of
the plans.
Volumetric Modulated Arc Therapy Created with
Machine Learning Versus Created Manually
Both VMAT (ML) plans and VMAT (manual)
plans demonstrated clinically acceptable treatment
plans. VMAT (manual) showed a significantly better
performance for target coverage of the breast or chest
wall and the nodal volumes [p < 0.001 for both PTV
(IMN) and PTV (axillary levels 3 and 4); p = 0.034 for
PTV (whole breast/chest wall)]. VMAT (ML) plans had significantly lower mean doses to the contralateral lung
and the heart compared to VMAT (manual) [p = 0.027
and p = 0.009, respectively] (Table 7). The time required
for treatment planning of a VMAT (ML) plan was
11.65 ± 2.00 minutes, while that of a VMAT (manual)
plan was 15.42 ± 5.24 minutes (p = 0.023).
Intensity Modulated Radiation Therapy Created
with Machine Learning Versus Created Manually
IMRT (ML) plans and VMAT (manual) plans had
comparable performance in terms of target coverage for
the breast or chest wall and PTV (axillary levels 3 and 4);
however, IMRT (ML) has a higher coverage of the PTV
(IMN) than IMRT (manual) [99.06% vs. 96.96%; p = 0.013]. For OARs, IMRT (ML) plans had a higher dose to the ipsilateral lung in terms of both mean dose and
V17Gy (both p < 0.001). The mean dose to ipsilateral lung
(14.81 ± 1.26 Gy) failed to fulfil the dose constraint. The
differences in mean dose and V17Gy to the heart were not
statistically significant (Table 8). The time required for
treatment planning of an IMRT (ML) plan was 51.47 ±
12.46 minutes, while that of a IMRT (manual) plan was
30.84 ± 5.20 minutes (p < 0.001).
DISCUSSION
Contouring
AI has been revolutionising various sectors, including
the medical field, impacting the system from daily
patient care to policy formulation.[27] [28] The advancements
in AI have significantly enhanced radiological imaging through improved image recognition using deep learning
algorithms. The clinical utilisation of AI in radiotherapy
delivery is on the rise, encompassing tasks such as
segmentation and treatment planning, in malignancies
arising from different regions including head and neck,
thorax and pelvis.[29] The primary goals are to enhance quality, decrease inter-observer variability, standardise
contouring, and increase efficiency.
Based on our study findings, AI demonstrated
encouraging results in segmentation of OARs. The
results of 95% HD and vDSC for heart and lungs were comparable to those of other AI models.[10] [30] This
achievement can be attributed to the clear visualisation
and differentiation of the heart and lungs, which also
exhibit minimal variability across different patients.
For clinical target volume, favourable performance was
observed for axillary level 2 and interpectoral nodes.
Delineation of these structures is closely associated with
identification of the pectoralis minor muscle, which is a
readily identified structure on CT, thus contributing to
improved consistency in contouring.
The deep learning segmentation model employed in our
study exhibited limitations in segmentation in some of
the patients, failing to delineate axillary level 1 in two
patients (7.4%) and the IMN in four patients (14.8%).
In addition, the model’s inability to identify the chest
wall underscores its constraints in clinical application.
The agreement between auto-segmentation and manual
contouring was found to be less consistent for axillary
level 1 compared to axillary levels 2 to 4, with larger 95%
HD and smaller sDSC. From the analysis of SLNB and
AD patients, better outcomes were more likely in those
who underwent SLNB instead of AD, despite the small
number of SLNB cases. Contouring of axillary level 1
is typically influenced by axillary surgery to encompass
the postoperative OARs or areas with surgical clips
based on clinical judgement rather than solely relying on
anatomical structures, leading to possible inter-observer
variability. The results suggest that disrupted anatomy
may also pose a challenge for auto-segmentation.
Apart from individual factors, variations between
manual contouring and auto-segmentation can also be
attributed to systemic factors. For example, patients
from our institute may have had different physiques
compared to the patients recruited in the AI training
dataset. Additionally, the quality of the CT imaging can
be a contributing factor, as it plays a significant role in the
visibility of structures such as the axillary and subclavian
vessels. These structures are crucial for defining the
boundaries of the axillary levels and consequently
influence the precision of target volume delineation by
both manual and auto-contouring.
Regarding the time spent for auto-segmentation and
the potential of AI to improve clinical efficiency, in
our study, the mean duration for auto-segmentation
was found to be <30 seconds. Although the time spent
on manual contouring was not documented, clinical
experience from our institute and existing literature[31] [32]
suggest that it typically requires approximately 35 minutes for manual contouring of a breast radiotherapy
plan, which is significantly longer than the time required
by AI.
At present, although vDSC with 0.7 indicates excellent
agreement in general,[25] there are no established
acceptable thresholds for the four metrics for clinical
use. In this study, the metrics provided only objective
numerical analysis, without incorporating qualitative
review by oncologists to gauge user satisfaction. This is a
limitation of this study, as the ultimate goal of reviewing
performance of AI in this domain is to augment manual
processes, where user acceptance is crucial for clinical
implementation of the technology.[33] Additionally, the
small number of oncologists reviewing the manual
contours as ground truth may not adequately represent
the intra- and inter-observer variability in reality. This
could be better addressed by creating a consensus manual
contour developed by a larger group of oncologists.
Another limitation of this study is that, while the findings
indicate that auto-segmentation requires significantly
less time than manual contouring, further assessment
of the time needed for manual review and correction of
auto-segmentation is necessary to fully evaluate its time-saving
benefits. Surface DSC is a relatively novel metric
for assessing the performance of auto-segmentation and
has been demonstrated to be a more effective indicator
of correction time compared to traditional metrics,
such as vDSC.[23] [24] Generally, it is anticipated that no
time savings will be realised if an observer expects to
alter an automatically generated contour by more than
approximately 40%.[23] Future research is needed to
investigate the relationship between surface DSC and
other relevant metrics with time savings, and establish
acceptable thresholds for clinical practice.
Treatment Planning
Adjuvant radiotherapy for breast cancer has been
associated with long-term complications. One notable
concern is the increased risk of cardiac toxicity,[34] and the
risk is particularly pronounced in women with existing
cardiac risk factors, or women receiving radiotherapy for
left-sided breast cancer. Additionally, there is a risk of
pneumonitis[35] and the development of second primary
cancers.[36] Although a recent meta-analysis found no
significant differences in the occurrence of cardiac
events and pneumonitis between patients who received
IMN irradiation and those who did not,[37] it is still
recommended to prioritise minimising the radiation dose
to OARs as much as possible, following the ALARA (As Low As Reasonably Achievable) principle.[38]
Facing the expected growing numbers of patients
undergoing radiotherapy including the IMN, a shift from
three-dimensional conformal radiation therapy to inverse
planning is needed to improve the quality of radiotherapy
plan. As it is likely to impose a substantial increase in the
workload associated with treatment planning, there is a
need to see if AI can facilitate the radiotherapy planning
process. The goal is to carry out treatment planning
efficiently while maintaining established standards.
The virtual plans in this study, including VMAT (ML),
VMAT (manual), IMRT (ML), and IMRT (manual),
were created based on contouring performed by
oncologists, and all of them demonstrated satisfactory
target coverage. VMAT (ML), VMAT (manual)
and IMRT (manual) successfully met all of the dose
constraints, while IMRT (ML) exceeded the objective
for the ipsilateral lung mean dose, which was measured
at 14.81 ± 1.26 Gy, surpassing the desired limit of ≤13 Gy.
In the evaluation of VMAT plans, VMAT (manual)
demonstrated superior target coverage compared to
VMAT (ML) plans, which exhibited a trend towards
reduced doses to OARs. Both planning approaches,
however, are deemed acceptable from a dosimetric
perspective.
In the context of IMRT plans, IMRT (manual) is regarded
as more acceptable, as it successfully meets the specified
dose objectives and constraints. In contrast, IMRT (ML)
will require manual adjustments and optimisation before
clinical application.
In our study, the time required to generate a VMAT
(ML) plan was faster by 3.77 minutes compared to
VMAT (manual) plan. The time taken to create an IMRT
(manual) plan was faster than the IMRT (ML) plan by
20.63 minutes. Notably, during the AI planning process
for both IMRT and VMAT, planners can attend to other
responsibilities while the system generates the treatment
plans; thus, the actual clinical time dedicated to AI
planning may be less than the recorded duration. On
the other hand, it is also important to acknowledge that
AI-generated plans are not inherently perfect and may
necessitate further adjustments to tailor the treatment
for individual patients. A limitation of this study is that
it does not quantify the time required for optimising an
AI-generated plan after its initial creation. These factors may influence the actual clinical time saving benefits
derived from the use of AI in treatment planning.
CONCLUSION
In the first part of the study, AI has demonstrated
reliable and excellent performance in auto-segmentation
of OARs including heart and lungs for breast cancer
radiotherapy. While the technology may also be applied
to the delineation of the breast and regional nodal targets,
it is crucial to bear in mind the need for case-by-case
adjustment, particularly for postoperative changes.
In the second part, the study showed that satisfactory
VMAT plans can be efficiently generated using an
automated approach, achieving performance comparable
to that of manual planning in a relatively short timeframe.
This method holds potential for clinical application,
warranting further investigation into its impact on
improving workflow in radiotherapy practices.
REFERENCES
1. World Health Organization. Breast cancer. 2024 Mar 13. Available
from: https://www.who.int/news-room/fact-sheets/detail/breast-cancer. Accessed 2 Jul 2024.
2. Centre for Health Protection, Department of Health, Hong Kong
SAR Government. Breast cancer. 2025 Feb 24. Available from:
https://www.chp.gov.hk/en/healthtopics/content/25/53.html.
Accessed 5 Mar 2025.
3. Thorsen LB, Overgaard J, Matthiessen LW, Berg M, Stenbygaard L,
Pedersen AN, et al. Internal mammary node irradiation in patients
with node-positive early breast cancer: fifteen-year results from
the Danish Breast Cancer Group Internal Mammary Node study.
J Clin Oncol. 2022;40:4198-206. Crossref
4. Boekel NB, Schaapveld M, Gietema JA, Russell NS, Poortmans P,
Theuws JC, et al. Cardiovascular disease risk in a large, population-based cohort of breast cancer survivors. Int J Radiat Oncol Biol
Phys. 2016;94:1061-72. Crossref
5. Chargari C, Castadot P, Macdermed D, Vandekerkhove C,
Bourgois N, Van Houtte P, et al. Internal mammary lymph node
irradiation contributes to heart dose in breast cancer. Med Dosim.
2010;35:163-8. Crossref
6. Siddique S, Chow JC. Artificial intelligence in radiotherapy. Rep
Prac Oncol Radiother. 2020;25:656-66. Crossref
7. Francolini G, Desideri I, Stocchi G, Salvestrini V, Ciccone LP,
Garlatti P, et al. Artificial intelligence in radiotherapy: state of the
art and future directions. Med Oncol. 2020;37:50. Crossref
8. Men K, Zhang T, Chen X, Chen B, Tang Y, Wang S, et al. Fully
automatic and robust segmentation of the clinical target volume
for radiotherapy of breast cancer using big data and deep learning.
Phys Med. 2018;50:13-9. Crossref
9. Liu Z, Liu F, Chen W, Tao Y, Liu X, Zhang F, et al. Automatic
segmentation of clinical target volume and organs-at-risk for breast
conservative radiotherapy using a convolutional neural network.
Cancer Manag Res. 2021;13:8209-17. Crossref
10. Almberg SS, Lervåg C, Frengen J, Eidem M, Abramova TM,
Nordstrand CS, et al. Training, validation, and clinical
implementation of a deep-learning segmentation model for
radiotherapy of loco-regional breast cancer. Radiother Oncol. 2022:173:62-8. Crossref
11. van de Sande D, Sharabiani M, Bluemink H, Kneepkens E, Bakx N,
Hagelaar E, et al. Artificial intelligence–based treatment planning
of radiotherapy for locally advanced breast cancer. Phys Imaging
Radiat Oncol. 2021;20:111-6. Crossref
12. Kneepkens E, Bakx N, van der Sangen M, Theuws J, van der
Toorn PP, Rijkaart D, et al. Clinical evaluation of two AI models for
automated breast cancer plan generation. Radiat Oncol. 2022;17:25. Crossref
13. American Joint Committee on Cancer. AJCC Stating Manual Eight
Edition. 2018. Available from: http://www.breastsurgeonsweb.com/wp-content/uploads/downloads/2020/10/AJCC-Breast-Cancer-Staging-System.pdf. Accessed 4 Mar 2025.
14. Varian. Eclipse. Available from: https://www.varian.com/products/radiotherapy/treatment-planning/eclipse. Accessed 2 Jul 2024.
15. RaySearch Laboratories. RayStation. Available from: https://www.raysearchlabs.com/raystation/. Accessed 2 Jul 2024.
16. Offersen BV, Boersma LJ, Kirkove C, Hol S, Aznar MC, Biete Sola A,
et al. ESTRO consensus guideline on target volume delineation for
elective radiation therapy of early-stage breast cancer. Radiother
Oncol. 2015;114:3-10. Crossref
17. The Royal College of Radiologists. Postoperative radiotherapy for
breast cancer: UK consensus statements. November 2016. Available
from: https://www.rcr.ac.uk/our-services/all-our-publications/clinical-oncology-publications/postoperative-radiotherapy-for-breast-cancer-uk-consensus-statements/. Accessed 2 Jul 2024.
18. Danish Breast Cancer Cooperative Group. The SKAGEN Trial 1.
Moderately hypofractionated loco-regional adjuvant radiation
therapy of early breast cancer combined with a simultaneous
integrated boost in patients with an indication for boost: DBCG
HYPO II, a randomised clinically controlled trial. 2015. Available
from: https://www.dbcg.dk/PDF%20Filer/SKAGEN%20Trial%201_%20protokol.pdf. Accessed 2 Jul 2024.
19. Mackay K, Bernstein D, Glocker B, Kamnitsas K, Taylor A. A
review of the metrics used to assess auto-contouring systems in
radiotherapy. Clin Oncol (R Coll Radiol). 2023;35:354-69. Crossref
20. Huttenlocher DP, Klanderman GA, Rucklidge WJ. Comparing
images using the Hausdorff distance. IEEE Trans Pattern Anal
Mach Intell. 1993;15:850-63. Crossref
21. Nikolov S, Blackwell S, Zverovitch A, Mendes R, Livne M,
De Fauw J, et al. Clinically applicable segmentation of head and neck
anatomy for radiotherapy: deep learning algorithm development and
validation study. J Med Internet Res. 2021;23:e26151. Crossref
22. Doolan PJ, Charalambous S, Roussakis Y, Leczynski A, Peratikou M,
Benjamin M, et al. A clinical evaluation of the performance of
five commercial artificial intelligence contouring systems for
radiotherapy. Front Oncol. 2023;13:1213068. Crossref
23. Vaassen F, Hazelaar C, Vaniqui A, Gooding M, van der Heyden B,
Canters R, et al. Evaluation of measures for assessing time-saving
of automatic organ-at-risk segmentation in radiotherapy. Phys
Imaging Radiat Oncol. 2019;13:1-6. Crossref
24. Kiser KJ, Barman A, Stieb S, Fuller CD, Giancardo L. Novel
autosegmentation spatial similarity metrics capture the time
required to correct segmentations better than traditional metrics
in a thoracic cavity segmentation workflow. J Digit Imaging.
2021;34:541-53. Crossref
25. Zijdenbos AP, Dawant BM, Margolin RA, Palmer AC.
Morphometric analysis of white matter lesions in MR images:
method and validation. IEEE Trans Med Imaging. 1994;13:716-24. Crossref
26. Bartko JJ. Measurement and reliability: statistical thinking
considerations. Schizophr Bull. 1991;17:483-9. Crossref
27. Ramezani M, Takian A, Bakhtiari A, Rabiee HR, Ghazanfari S,
Mostafavi H. The application of artificial intelligence in health
policy: a scoping review. BMC Health Serv Res. 2023;23:1416. Crossref
28. Amisha, Malik P, Pathania M, Rathaur VK. Overview of artificial
intelligence in medicine. J Family Med Prim Care. 2019;8:2328-31. Crossref
29. Wang C, Zhu X, Hong JC, Zheng D. Artificial intelligence in
radiotherapy treatment planning: present and future. Technol
Cancer Res Treat. 2019;18:153303381987392. Crossref
30. Matoska T, Patel M, Liu H, Beriwal S. Review of deep learning
based autosegmentation for clinical target volume: current status
and future directions. Adv Radiat Oncol. 2024;9:101470. Crossref
31. Andrianarison VA, Laouiti M, Fargier-Bochaton O, Dipasquale G,
Wang X, Nguyen NP, et al. Contouring workload in adjuvant breast
cancer radiotherapy. Cancer Radiother. 2018;22:747-53. Crossref
32. Bakx N, Rijkaart D, van der Sangen M, Theuws J, van der Toorn PP,
Verrijssen AS, et al. Clinical evaluation of a deep learning
segmentation model including manual adjustments afterwards for
locally advanced breast cancer. Tech Innov Patient Support Radiat
Oncol. 2023;26:100211. Crossref
33. Buelens P, Willems S, Vandewinckele L, Crijns W, Maes F,
Weltens CG. Clinical evaluation of a deep learning model for
segmentation of target volumes in breast cancer radiotherapy.
Radiother Oncol. 2022;171:84-90. Crossref
34. Bradshaw PT, Stevens J, Khankari N, Teitelbaum SL, Neugut AI,
Gammon MD. Cardiovascular disease mortality among breast
cancer survivors. Epidemiology. 2016;27:6-13. Crossref
35. Lingos TI, Recht A, Vicini F, Abner A, Silver B, Harris JR.
Radiation pneumonitis in breast cancer patients treated with
conservative surgery and radiation therapy. Int J Radiat Oncol Biol
Phys. 1991;21:355-60. Crossref
36. Stovall M, Smith SA, Langholz BM, Boice JD Jr, Shore RE,
Andersson M, et al. Dose to the contralateral breast from
radiotherapy and risk of second primary breast cancer in the
WECARE study. Int J Radiat Oncol Biol Phys. 2008;72:1021-30. Crossref
37. Shaikh PM, Mulherkar R, Khasawneh MT, Clump D, Hazard-Jenkins H, Hafez M, et al. Treatment of internal mammary nodes is associated with improved overall survival in breast cancer: a
meta-analysis. Am J Clin Oncol. 2024;47:81-7. Crossref
38. Hendee WR, Edwards FM. ALARA and an integrated approach to
radiation protection. Semin Nucl Med. 1986;16:142-50. Crossref