Management and Prognosis of Breast Intraductal Papilloma Diagnosed by Core Needle Biopsy: Comparing Vacuum-assisted Excision, Surgical Excision, and Surveillance
ORIGINAL ARTICLE
Hong Kong J Radiol 2025 Mar;28(1):e14-22 | Epub 17 March 2025
Management and Prognosis of Breast Intraductal Papilloma Diagnosed by Core Needle Biopsy: Comparing Vacuum-assisted
Excision, Surgical Excision, and Surveillance
HL Chan1, KH Wong1, KF Tam1, HHL Chau2
1 Department of Radiology, North District Hospital, Hong Kong SAR, China
2 Department of Imaging and Interventional Radiology, Prince of Wales Hospital, Hong Kong SAR, China
Correspondence: Dr HL Chan, Department of Radiology, North District Hospital, Hong Kong SAR, China. Email: hollischanrad@gmail.com
Submitted: 20 June 2023; Accepted: 8 November 2023.
Contributors: All authors designed the study. HLC and KHW acquired the data. All authors analysed the data. HLC and KHW drafted the
manuscript. KFT and HHLC critically revised the manuscript for important intellectual content. All authors had full access to the data,
contributed to the study, approved the final version for publication, and take responsibility for its accuracy and integrity.
Conflicts of Interest: All authors have disclosed no conflicts of interest.
Funding/Support: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data Availability: All data generated or analysed during the present study are available from the corresponding author on reasonable request.
Ethics Approval: This research was approved by the Joint Chinese University of Hong Kong–New Territories East Cluster Clinical Research Ethics Committee, Hong Kong (Ref No.: 2023.169). The requirement for patient consent was waived by the Committee due to the retrospective nature of the research.
Abstract
Introduction
Intraductal papilloma (IDP) is a common breast lesion that is often excised, but the treatment of
choice for IDP without atypia diagnosed on core needle biopsy (CNB) remains controversial due to its low risk of
malignancy. Besides surgery, vacuum-assisted excision (VAE) has emerged as a less invasive alternative. This study
aimed to compare the management and prognosis of IDP without atypia diagnosed by CNB across surgical excision,
VAE and surveillance, and to identify risk factors predicting papilloma malignant transformation.
Methods
This single-centre retrospective review included 107 consecutive samples diagnosed with IDP without
atypia with ultrasound-guided CNB from 2016 to 2020. The patients underwent surgical excision, ultrasound-guided
VAE or surveillance. The malignant transformation and recurrence rates were evaluated, and potential risk factors
for malignant transformation were analysed.
Results
The overall malignant transformation rate was 3.7%. The malignant transformation rates were 10.3% in
the surgical excision group and 2.1% in the VAE group. No IDP recurrence was identified in either group. For the
group that underwent surveillance, none of the lesions showed significant size increase during follow-up. Lesions
≤1 cm and nonpalpable lesions showed low malignant transformation rates of 1.3% and 1.2%, respectively. Risk
factors for malignant transformation included larger size (p = 0.002), palpability (p = 0.016), multiple lesions (p = 0.045), and a positive family history of breast cancer (p = 0.035).
Conclusion
Imaging surveillance may be an alternative management option for IDP in low-risk groups given its
low malignant transformation rate. VAE is a safe and effective choice. Surgery may be considered for larger sized
lesions with risk factors.
Key Words: Biopsy, large-core needle; Papilloma, intraductal; Radiology information systems
中文摘要
芯針活檢診斷乳腺導管內乳頭狀瘤的治療和預後:比較真空輔助切除、手術切除和非外科監測
陳凱玲、黃健開、譚國輝、周海倫
引言
導管內乳頭狀瘤是一種常見的乳腺病變,通常外科切除,但由於其惡性風險較低,對於經芯針活檢診斷無異型增生導管內乳頭狀瘤的治療選擇仍存在爭議。除了手術之外,真空輔助切除成為創傷較小的替代方案。本研究旨在比較芯針活檢診斷的無異型增生導管內乳頭狀瘤的手術切除、真空輔助切除和非外科監測的治療和預後,並找出預測乳頭狀瘤惡變的風險因素。
方法
本單中心回顧性研究納入了2016至2020年間經超音波引導下的芯針活檢診斷為無異型增生導管內乳頭狀瘤的107例連續樣本。患者接受了手術切除、超音波引導下的真空輔助切除或非外科監測。我們評估了惡變和復發率,並分析了惡變的潛在風險因素。
結果
整體惡變率為3.7%。手術切除組的惡變率為10.3%,而真空輔助切除組的惡變率為2.1%。兩組均未發現導管內乳頭狀瘤復發。接受非外科監測組在隨訪期間沒有病變顯示出明顯大小增加。≤1 cm 的病灶和不可觸及病灶的惡變率低,分別為1.3%和1.2%。惡變的風險因素包括體積較大(p = 0.002)、可觸及(p = 0.016)、多發性病灶(p = 0.045)和乳癌陽性家族史(p = 0.035)。
結論
鑑於影像監測的惡變率低,因此它可以是治療低風險群導管內乳頭狀瘤的替代方案。真空輔助切除是安全有效的選擇。具有惡變風險因素的較大病變可以考慮外科手術。
INTRODUCTION
Intraductal papillomas (IDPs) of the breast are common
benign lesions that arise from the epithelium lining the
lactiferous ducts. They can be found incidentally during
mammography, ultrasonography (US), or magnetic
resonance imaging, or present with symptoms such as
nipple discharge, palpable masses, or asymptomatic.
IDPs can be classified as solitary or multiple, and with
or without atypia. IDPs without atypia are benign lesions
with a low risk of developing into breast cancer.[1] [2] [3]
However, IDPs with atypia have a higher risk of
malignant transformation. Consensus exists regarding the
need for surgical excision of IDP with atypia diagnosed
on core needle biopsy (CNB), owing to a pooled
malignant transformation rate of up to 36.9%.[4] However,
despite numerous published studies, the management of
IDP without atypia diagnosed by CNB is still a matter
of debate.[5] Surgical excision has been the standard
approach for many years, with the goal of establishing
a definitive diagnosis, excluding coexisting malignancy,
and preventing progression to cancer. It is the most
commonly used management strategy, but it has some
drawbacks, such as the need for general anaesthesia,
potential complications, and possible deformity. In recent
years, vacuum-assisted excision (VAE) has emerged as an alternative to surgical excision, as it is less invasive,
more convenient, and associated with lower morbidity.[6]
Follow-up breast imaging has also been proposed as
an alternative to excisional management in selected
patients, with the advantage of avoiding the risks and
complications associated with surgery.[7]
Studies have reported the malignant transformation
rate and the recurrence rate after surgical excision or
VAE of IDP without atypia.[1] [7] However, there is still
no consensus on the best management strategy for this
lesion, and some studies have suggested that follow-up
breast imaging may be a safe and effective management
strategy in selected patients.[3] [4] [7] The aim of this study was
to estimate the feasibility of VAE or surveillance for the
management of IDP without atypia compared to surgical
excision, with regard to malignant transformation rate
and the recurrence rate. The potential risk factors for
malignant transformation were also analysed.
METHODS
We conducted a retrospective analysis of 173 consecutive
biopsy specimens of IDP without atypia obtained with
CNB under US guidance at our centre between 1 January
2016 and 31 December 2020. We excluded patients with ipsilateral breast cancer who underwent total mastectomy
and patients lost to follow-up. For multiple IDPs
diagnosed on CNB in the same patient, only the largest
IDP was included in the study and the rest were excluded.
The management strategy for each patient was
determined in multidisciplinary team meetings with
breast radiologists, breast surgeons, and pathologists.
The management options included surgical excision,
VAE, and surveillance by follow-up breast US. During
the multidisciplinary meetings, pathologists meticulously
reviewed the tissue slices for any atypical features and
the adequacy of core samples for diagnostic confidence.
Radiologists analysed the ultrasound images to establish
concordance between radiological and pathological
findings, making sure no possible malignancy was
present. Additionally, the feasibility of surgery or
VAE was assessed based on factors such as lesion size,
location (peripheral or central) and depth from the skin.
Multidisciplinary team members also identified potential
risk factors associated with malignant transformation,
including patient demographics, family history of
breast cancer, larger lesion size, and related symptoms.
Most importantly, patientsʼ willingness to proceed to
lesion removal or preference of imaging surveillance
was addressed. Ultimately, the decision to pursue
imaging surveillance, surgery or VAE was guided by a
multidisciplinary evaluation of risks and benefits, and
most importantly respecting the patient’s wishes.
For patients in the surgical excision group, the surgical
excision was performed by the surgical team, and the
information was extracted from the surgical record. For
patients in the imaging surveillance group, the size and
location of each lesion were recorded at the time of initial
diagnosis and latest follow-up.
For patients in the VAE group, the procedure was
performed by radiologists in our department. The
technique of US-guided VAE is illustrated in Figures 1, 2 and 3. The targeted lesion, which was previously proven
IDP without atypia on CNB, would first be identified on
preliminary US. Infusion to the skin with 1 to 5 mL of 2%
lignocaine and the perilesional region with 5 to 10 mL
1:200 000 adrenaline was used as local anaesthesia. The
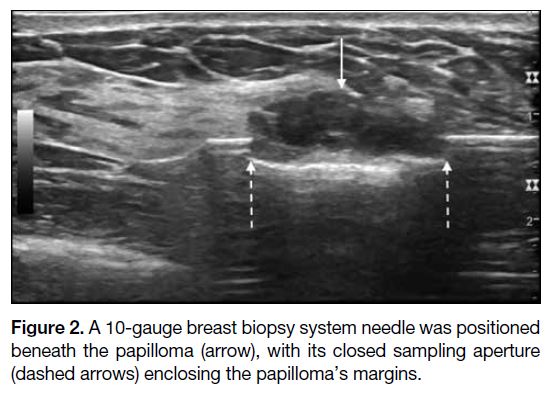
probe was introduced through a small skin incision and
the IDP was positioned within the margins of the closed
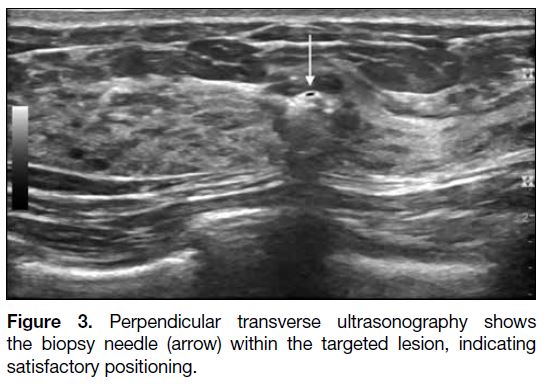
sampling aperture (Figure 2) and satisfactory positioning
was confirmed on imaging in a plane perpendicular to
the first image (Figure 3). In our institute, we used a
10-gauge EnCor Enspire Breast Biopsy System needle
(SenoRx, Aliso Viejo [CA], United States). Samples
were taken with adjustment (rotation and repositioning)
of the needle, if necessary, until the papilloma was
indiscernible on US.
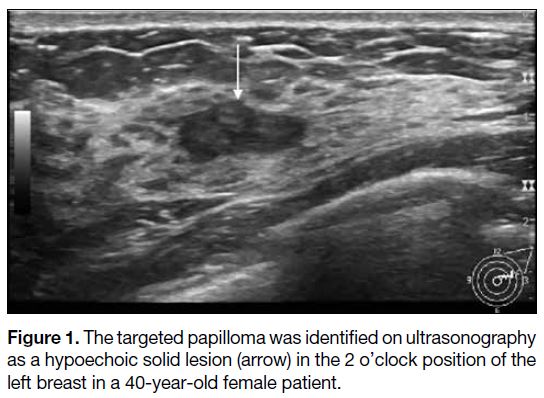
Figure 1. The targeted papilloma was identified on ultrasonography
as a hypoechoic solid lesion (arrow) in the 2 o’clock position of the
left breast in a 40-year-old female patient.
Figure 2. A 10-gauge breast biopsy system needle was positioned
beneath the papilloma (arrow), with its closed sampling aperture
(dashed arrows) enclosing the papilloma’s margins.
Figure 3. Perpendicular transverse ultrasonography shows
the biopsy needle (arrow) within the targeted lesion, indicating
satisfactory positioning.
Patients’ demographics and clinical data, radiological
data, histopathological diagnosis, malignant
transformation rates, and recurrence rates were recorded (Table 1). These data were extracted from the electronic
health record and the radiology information system.
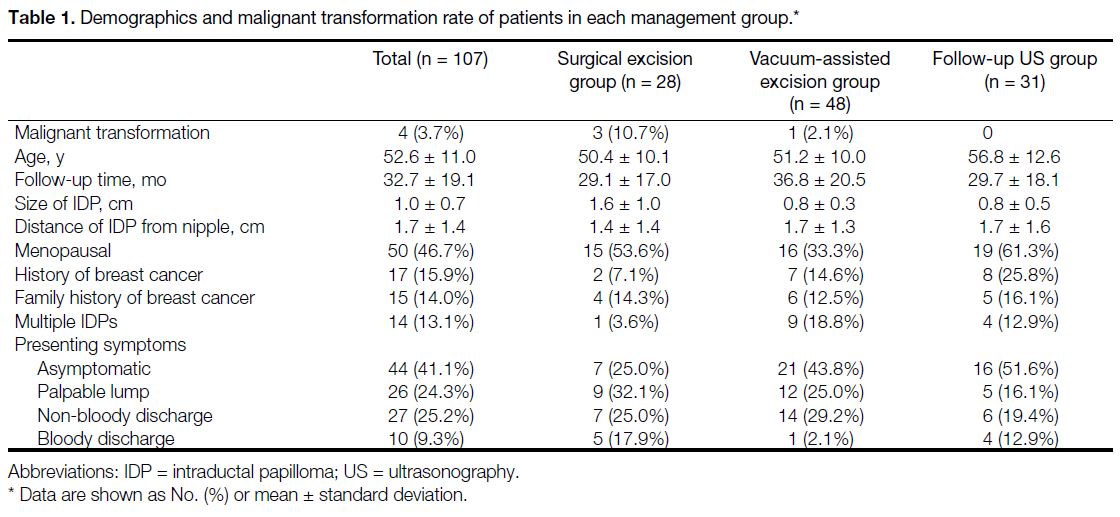
Table 1. Demographics and malignant transformation rate of patients in each management group.
On ultrasound, IDPs typically present as solid or
complex cystic and solid masses. It is also possible to
identify the lesion within a dilated duct in some cases,
as demonstrated in Figure 1. The size of the IDP was
measured as the longest dimension on ultrasound, and
the location of the lesion was measured as the distance
from the nipple (in cm). The malignant transformation
rate was defined as the percentage of patients who were
diagnosed with malignancy on surgical excision or VAE
after the initial diagnosis of IDP without atypia. The
recurrence rate was defined as the percentage of patients
who were found to have evidence of recurrence (i.e., the
rate of returning to previous histological grade) of the
lesion on follow-up imaging after surgical excision or
VAE. A patient was considered to have multiple IDPs
if there was a previous history of IDP, or if multiple
biopsies had been performed at different sites within 60
days diagnosing IDPs.
A previous personal history of breast cancer included a
history of invasive disease or ductal carcinoma in situ.
The follow-up period was defined as the time from the
initial diagnosis to the last imaging study.
Statistical Analyses
Statistical analyses were performed using commercial
software SPSS (Windows version 26.0; IBM Corp,
Armonk [NY], United States). Categorical variables
were expressed as frequencies and percentages, and continuous variables were expressed as means ±
standard deviations (SDs). The Pearson Chi squared test
or Fisher’s exact test was used to compare the categorical
variables, and the independent t test or Mann-Whitney
U test was used to compare the continuous variables
between groups. A p value of < 0.05 was considered
significant.
RESULTS
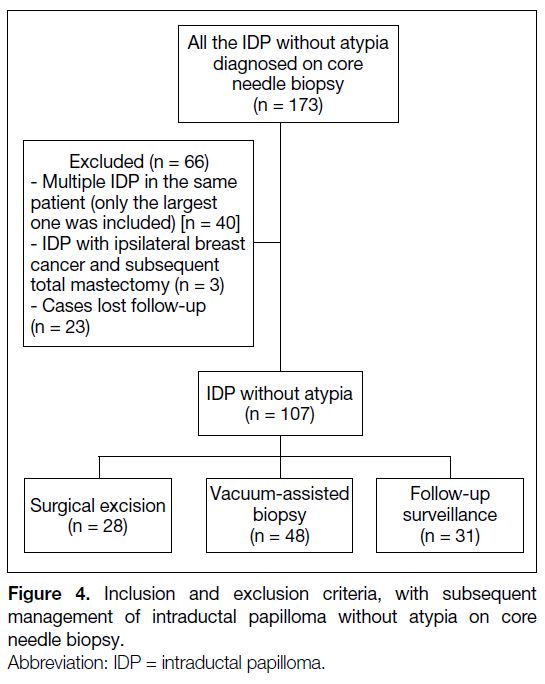
The 173 consecutive samples with pathological
diagnosis of IDP without atypia in 2016 to 2020 were
reviewed. After excluding lesions except the largest one
in one patient with multiple IDPs (n = 40), patients with
ipsilateral breast cancer with total mastectomy (n = 3)
and patients who were lost to follow-up (n = 23), we
included 107 patients for subsequent analysis (Figure 4).
Figure 4. Inclusion and exclusion criteria, with subsequent
management of intraductal papilloma without atypia on core
needle biopsy.
Malignant Transformation Rate
The demographics and malignant transformation rates
of the patients are shown in Table 1. The mean age of
the study population was 52.6 years (SD = 11.0, range = 29-75) and the mean follow-up time was 32.7 months
(SD = 19.1, range = 3-77). All patients were female.
Of the 107 patients in the study, four (3.7%) were
diagnosed with malignancy after excision, for a
malignant transformation rate in the surgical excision
group of 10.7% and 2.1% in the VAE group. There was
no recurrence identified in either group. For the group
that underwent follow-up breast imaging, none of the
lesions showed increase in size during a mean follow-up
of 29.7 months (SD = 18.1, range = 5-63).
The mean size of the lesions in the surgical excision
group was larger (1.6 cm) compared to the VAE group
(0.8 cm) and the breast imaging follow-up group
(0.8 cm). The distance of the lesion from the nipple was
similar in all three groups (1.4 cm in the surgical excision
group, 1.7 cm in the VAE group, and 1.7 cm in the breast
imaging follow-up group) [Table 1].
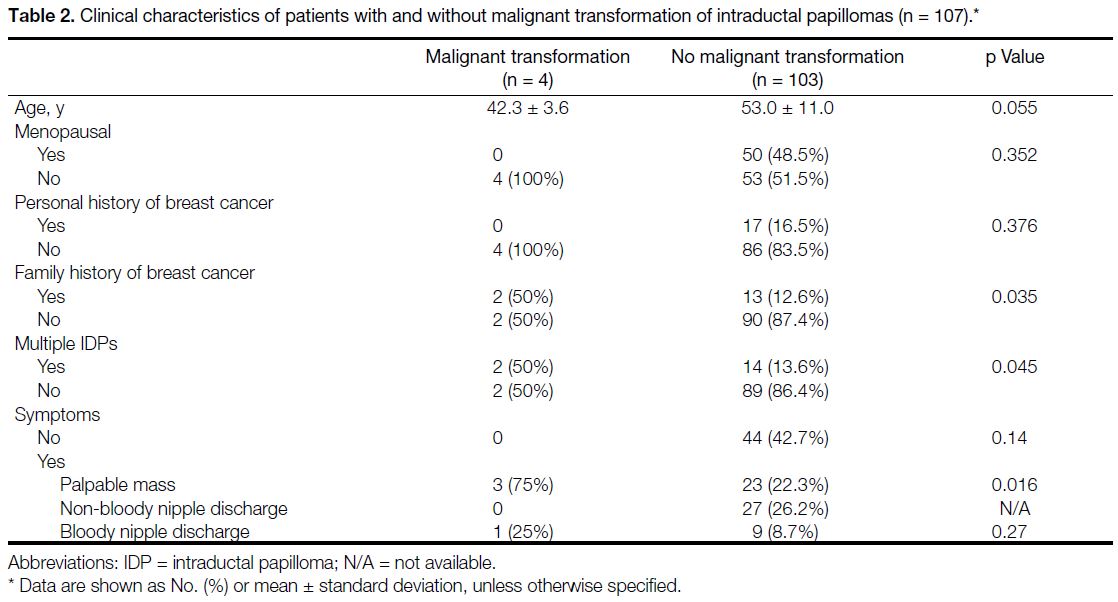
The clinical and lesion characteristics were analysed to
explore the potential association with histopathological
malignant transformation. The results of the marginal
analysis revealed that family history of breast cancer
(p = 0.035), presenting symptom of palpable mass
(p = 0.016), multiple IDPs (p = 0.045), and lesion
size (p = 0.002) were significantly associated with
malignant transformation (Tables 2 and 3). Lesions
≤1 cm and nonpalpable lesions showed low upgrade
rates (i.e., malignant transformation rates) of 1.33% and
1.23%, respectively (Table 4). Although the presenting
symptom of bloody nipple discharge was more frequent
in the malignant transformation group, it did not reach
statistical significance (Table 2). Similarly, lesions that
were located farther from the nipple were also found to
be more frequent in the malignant transformation group,
but this did not reach statistical significance either (p = 0.400). There were no significant differences in patients’ age (p = 0.055), menopausal status (p = 0.352),
or personal history of breast cancer (p = 0.376) among
the malignant transformation group versus the non-malignant
transformation group (Table 2).
Table 2. Clinical characteristics of patients with and without malignant transformation of intraductal papillomas (n = 107).
Table 3. Lesion characteristics of papilloma with and without malignant transformation (n = 107).
Table 4. Subgroup analysis of malignant transformation rates.
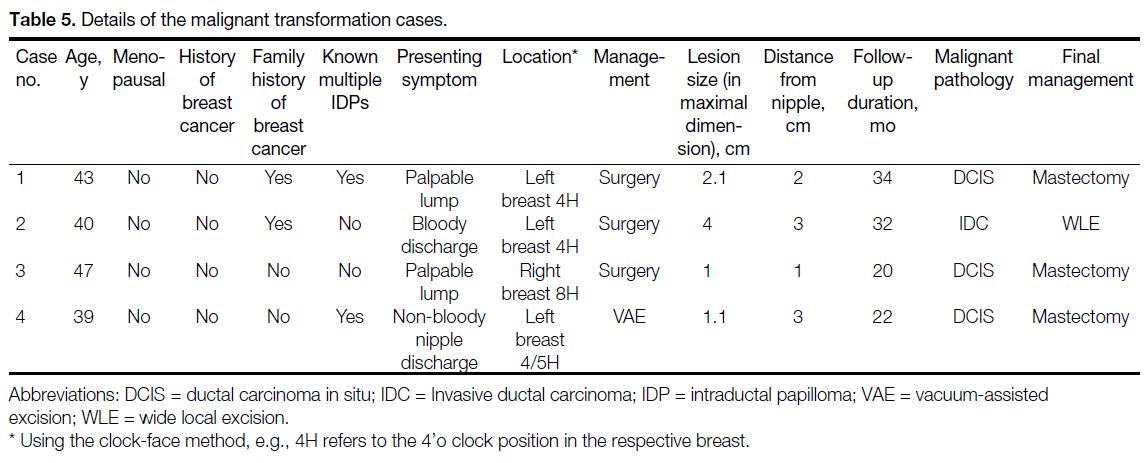
Of the four cases that underwent malignant
transformation, one was invasive ductal carcinoma and
the remaining three were ductal carcinoma in situ. Table 5 summarises the clinical and radiological details of these cases.
Table 5. Details of the malignant transformation cases.
Safety and Efficacy of Vacuum-assisted
Excision
During each VAE procedure, the lesion was removed
with real-time US guidance until it became completely
undetectable on US. The number of cores taken
depended on the size of the lesion and the position of the
VAE needle. The number of cores collected had a mean
of 8.43 per procedure (SD = 2.35).
Common complications of VAE include haematomas,
pain, and ecchymosis.[8] These are usually self-limiting
and do not require further medical attention and
management.[9] Any that may have developed after the
patient has left our department were not documented in
the radiological procedure report.
The procedure duration of VAE was documented
based on the time log on US capture images which
indicated the amount of time the radiologist spent for
the procedure, from the first captured US image to the
final image. The mean procedure time was 18 minutes
(SD = 6.44). Note that the documented procedure time did
not include post-procedure wound care, wound dressing,
or manual compression (routine postprocedural manual
compression of 10 minutes to ensure haemostasis).
DISCUSSION
Breast IDP without atypia can be difficult to diagnose
accurately; it is impossible to predict its malignant
transformation potential using radiological imaging
alone.[1] [10] [11] CNB is often used to obtain the diagnosis,
but there is controversy whether surgical excision is
necessary. In our study, we found that the malignant
transformation rate was 3.7% in the total study population,
consistent with published literature malignancy ranges
from 0 to 29%.[1] [2] [3] [12] [13] [14] [15] [16] [17]
Surgical excision has traditionally been the standard
management strategy for breast IDP,[18] [19] but VAE has
gained popularity in recent years due to its minimally invasive nature.[6] In our study, we found that there was
no recurrence of papillomas in patients undergoing
VAE with a mean follow-up period of 36.8 months
(SD = 20.5) and those undergoing surgical excision with
a mean follow-up period of 29.1 months (SD = 17.0)
[Table 1], suggesting that both strategies are effective in
preventing recurrence.
VAE is a more minimally invasive approach compared
with surgical excision. It has a shorter procedure time,
requiring only local anaesthesia, and results in minimal
scarring and a lower possibility of breast deformity.
Complication rates are very low and are self-limited.[20] Therefore, VAE is particularly suitable for low-risk
lesions and patients with cosmesis concern.
However, there are several drawbacks of VAE. First,
VAE removes the lesion in a ‘piecemeal’ fashion, which
precludes pathologists from assessing the margins of the
excision.[21] Also, the completeness of excision relies on
the sonographic appearance of the lesion. IDP can grow
into small branches of the ducts. There is difficulty in
differentiating adherent debris from IDP, especially
in peripheral ducts.[10] [22] Therefore, there is a potential
increased risk of residual lesional tissue.[23] Lastly, there
is a size limit for VAE, as larger sized lesions are
more technically difficult to excise, with a high risk of
haemorrhage and incomplete excision. Surgical excision
is preferred for lesions >2.5 cm.[24]
Our study also found that breast imaging follow-up
alone may be a reasonable management strategy for
selected patients with IDP without atypia. Breast IDPs
without atypia had a low malignant transformation rate of 3.7% in our study. However, the overall malignant
transformation rate in our study remained higher
than the 2% threshold in category 3 of the American
College of Radiology Breast Imaging Reporting and
Data System (BI-RADS).[25] This may make radiologists
reluctant to assign IDPs without atypia for follow-up
surveillance only. Nonetheless, for IDPs of sizes ≤1 cm
and nonpalpable lesions, the percentage of upgrade
to malignancy were 1.3% and 1.2% respectively,
justified to be classified as BI-RADS category 3 lesions.
Additionally, in our study, none of the patients in the
surveillance group experienced a significant increase
in lesion size or malignant transformation during a
mean follow-up of 29.7 months. This is also consistent
with reported literature.[26] [27] Therefore, we propose that
lesions with lower risk (≤1 cm or nonpalpable) and/or
without any risk factors (i.e., positive family history
of breast cancer and/or multiple IDPs) are justified to
undergo follow-up imaging surveillance in accord with
BI-RADS.
Research has been carried out to identify the possible
risk factors for malignant transformation. In our study,
we identified several associated factors in line with
previous studies, including lesion size,[26] [27] [28] [29] palpability,[26] [30]
multiplicity,[28] and a family history of breast cancer.[28]
Previous studies have suggested that older age correlates
with higher potential for malignant transformation.[14] [31] [32]
However, in our study, we could not demonstrate such
correlation.
Limitations
Our study has several limitations. First, it was a
retrospective study conducted at a single institution, which may limit the generalisability of our findings. Second, as
a retrospective study, the assignment of patients to each
management group was based on multiple factors rather
than randomisation, which resulted in selection bias and
therefore may be the reason for the different malignant
transformation rates in the three groups. Third, the sample
size was relatively small, which limits the statistical
power of our study. Meanwhile, the types of core biopsy
needle and the number of passes were not documented,
which may have affected the diagnostic accuracy of
CNB. Also, the follow-up period was relatively short,
and longer follow-up may be necessary to fully evaluate
the efficacy of each treatment approach. Lastly, as a
retrospective study, there was no established protocol
specified for IDP imaging follow-up. The follow-up
radiological examination appointment was assigned as
per clinical request. This would be a very important topic
for future study.
CONCLUSION
Our study confirmed low malignant transformation
and recurrence rates of IDP without atypia. Follow-up
imaging surveillance for low-risk lesions may be a
promising alternative management. We also suggest the
feasibility of using VAE instead of surgical excision
when clinically indicated, as a less invasive yet safe and
effective method of choice. Patients who undergo follow-up
with breast imaging should be closely monitored
for changes in the lesion size and should be advised to
undergo biopsy or excision if there is any suspicion of
malignancy. Surgery may be considered for larger or
palpable lesions, as well as for patients with multiple
IDPs or a positive family history of breast cancer, due
to their association with an increased risk of malignant transformation. Further studies with larger sample sizes
and longer follow-up periods are needed to validate our
findings.
REFERENCES
1. Pareja F, Corben AD, Brennan SB, Murray MP, Bowser ZL,
Jakate K, et al. Breast intraductal papillomas without atypia in
radiologic-pathologic concordant core-needle biopsies: rate of
upgrade to carcinoma at excision. Cancer. 2016;122:2819-27. Crossref
2. Maxwell AJ, Mataka G, Pearson JM. Benign papilloma diagnosed
on image-guided 14-G core biopsy of the breast: effect of lesion
type on likelihood of malignancy at excision. Clin Radiol.
2013;68:383-7. Crossref
3. Nayak A, Carkaci S, Gilcrease MZ, Liu P, Middleton LP,
Bassett RL Jr, et al. Benign papillomas without atypia diagnosed
on core needle biopsy: experience from a single institution and
proposed criteria for excision. Clin Breast Cancer. 2013;13:439-49. Crossref
4. Wen X, Cheng W. Nonmalignant breast papillary lesions at core-needle
biopsy: a meta-analysis of underestimation and influencing
factors. Ann Surg Oncol. 2013;20:94-101. Crossref
5. Dillon MF, McDermott EW, Hill AD, O’Doherty A, O’Higgins N,
Quinn CM. Predictive value of breast lesions of “uncertain
malignant potential” and “suspicious for malignancy” determined
by needle core biopsy. Ann Surg Oncol. 2007;14:704-11. Crossref
6. Wang ZL, Liu G, Huang Y, Wan WB, Li JL. Percutaneous
excisional biopsy of clinically benign breast lesions with vacuumassisted
system: comparison of three devices. Eur J Radiol.
2012;81:725-30. Crossref
7. Moynihan A, Quinn EM, Smith CS, Stokes M, Kell M, Barry JM,
et al. Benign breast papilloma: is surgical excision necessary?
Breast J. 2020;26:705-10. Crossref
8. Yoo HS, Kang WS, Pyo JS, Yoon J. Efficacy and safety of
vacuum-assisted excision for benign breast mass lesion: a metaanalysis.
Medicina (Kaunas). 2021;57:1260. Crossref
9. Wang WJ, Wang Q, Cai QP, Zhang JQ. Ultrasonographically
guided vacuum-assisted excision for multiple breast masses:
non-randomized comparison with conventional open excision. J
Surg Oncol. 2009;100:675-80. Crossref
10. Lam WW, Chu WC, Tang AP, Tse G, Ma TK. Role of radiologic
features in the management of papillary lesions of the breast. AJR
Am J Roentgenol. 2006;186:1322-7. Crossref
11. Shouhed D, Amersi FF, Spurrier R, Dang C, Astvatsaturyan K,
Bose S, et al. Intraductal papillary lesions of the breast: clinical
and pathological correlation. Am Surg. 2012;78:1161-5. Crossref
12. Ko D, Kang E, Park SY, Kim SM, Jang M, Yun BL, et al. The
management strategy of benign solitary intraductal papilloma on
breast core biopsy. Clin Breast Cancer. 2017;17:367-72. Crossref
13. Holley SO, Appleton CM, Farria DM, Reichert VC, Warrick J,
Allred DC, et al. Pathologic outcomes of nonmalignant papillary
breast lesions diagnosed at imaging-guided core needle biopsy.
Radiology. 2012;265:379-84. Crossref
14. Youk JH, Kim EK, Kwak JY, Son EJ, Park BW, Kim SI. Benign
papilloma without atypia diagnosed at US-guided 14-gauge core-needle
biopsy: clinical and US features predictive of upgrade to
malignancy. Radiology. 2011;258:81-8. Crossref
15. Tseng HS, Chen YL, Chen ST, Wu YC, Kuo SJ, Chen LS, et al.
The management of papillary lesion of the breast by core needle
biopsy. Eur J Surg Oncol. 2009;35:21-4. Crossref
16. Sohn YM, Park SH. Comparison of sonographically guided core
needle biopsy and excision in breast papillomas: clinical and
sonographic features predictive of malignancy. J Ultrasound Med. 2013;32:303-11. Crossref
17. Lee SJ, Wahab RA, Sobel LD, Zhang B, Brown AL, Lewis K, et al.
Analysis of 612 benign papillomas diagnosed at core biopsy: rate
of upgrade to malignancy, factors associated with upgrade, and a
proposal for selective surgical excision. AJR Am J Roentgenol.
2021;217:1299-311. Crossref
18. Chang JM, Moon WK, Cho N, Han W, Noh DY, Park IA, et al.
Management of ultrasonographically detected benign papillomas
of the breast at core needle biopsy. AJR Am J Roentgenol.
2011;196:723-9. Crossref
19. Puglisi F, Zuiani C, Bazzocchi M, Valent F, Aprile G, Pertoldi B,
et al. Role of mammography, ultrasound and large core biopsy in
the diagnostic evaluation of papillary breast lesions. Oncology.
2003;65:311-5. Crossref
20. Ko KH, Jung HK, Youk JH, Lee KP. Potential application of
ultrasound-guided vacuum-assisted excision (US-VAE) for wellselected
intraductal papillomas of the breast: single-institutional
experiences. Ann Surg Oncol. 2012;19:908-13. Crossref
21. Brzuszkiewicz K, Hodorowicz-Zaniewska D, Miękisz J,
Matyja A. Comparison of two minimally invasive biopsy
techniques—Breast Lesion Excision System and vacuum-assisted
biopsy—for diagnosing and treating breast lesions. Arch Med
Sci. 2022;18:1453-9. Crossref
22. Rissanen T, Reinikainen H, Apaja-Sarkkinen M. Breast
sonography in localizing the cause of nipple discharge:
comparison with galactography in 52 patients. J Ultrasound Med.
2007;26:1031-9. Crossref
23. Ueng SH, Mezzetti T, Tavassoli FA. Papillary neoplasms of the
breast: a review. Arch Pathol Lab Med. 2009;133:893-907. Crossref
24. Rageth CJ, O’Flynn EA, Pinker K, Kubik-Huch RA, Mundinger A,
Decker T, et al. Second International Consensus Conference on
lesions of uncertain malignant potential in the breast (B3 lesions).
Breast Cancer Res Treat. 2019;174:279-96. Crossref
25. Eberl MM, Fox CH, Edge SB, Carter CA, Mahoney MC. BI-RADS classification for management of abnormal mammograms.
J Am Board Fam Med. 2006;19:161-4. Crossref
26. Kuehner G, Darbinian J, Habel L, Axelsson K, Butler S, Chang S,
et al. Benign papillary breast mass lesions: favorable outcomes
with surgical excision or imaging surveillance. Ann Surg Oncol.
2019;26:1695-703. Crossref
27. Wyss P, Varga Z, Rössle M, Rageth CJ. Papillary lesions of the
breast: outcomes of 156 patients managed without excisional
biopsy. Breast J. 2014;20:394-401. Crossref
28. Liberman L, Tornos C, Huzjan R, Bartella L, Morris EA,
Dershaw DD. Is surgical excision warranted after benign,
concordant diagnosis of papilloma at percutaneous breast biopsy?
AJR Am J Roentgenol. 2006;186:1328-34. Crossref
29. MacColl C, Salehi A, Parpia S, Hodgson N, Ramonas M,
Williams P. Benign breast papillary lesions diagnosed on core
biopsy: upgrade rate and risk factors associated with malignancy
on surgical excision. Virchows Arch. 2019;475:701-7. Crossref
30. Jung SY, Kang HS, Kwon Y, Min SY, Kim EA, Ko KL, et al.
Risk factors for malignancy in benign papillomas of the breast
on core needle biopsy. World J Surg. 2010;34:261-5. Crossref
31. Rizzo M, Linebarger J, Lowe MC, Pan L, Gabram SG, Vasquez
L, et al. Management of papillary breast lesions diagnosed on
core-needle biopsy: clinical pathologic and radiologic analysis of
276 cases with surgical follow-up. J Am Coll Surg. 2012;214:280-7. Crossref
32. Glenn ME, Throckmorton AD, Thomison JB 3rd, Bienkowski RS.
Papillomas of the breast 15 mm or smaller: 4-year experience in
a community-based dedicated breast imaging clinic. Ann Surg
Oncol. 2015;22:1133-9. Crossref